Ozonolysis is the process by which ozone (O3) reacts with alkenes (olefins) to break the double bond and form two carbonyl groups. If the double bond of the alkene is substituted with hydrogen or carbon atoms, the carbonyl groups that are formed are either aldehydes or ketones. Acyclic alkenes form two carbonyl compounds while cyclic alkenes produce a single compound containing two carbonyl groups. As an analytical tool, ozonolysis reveals the substitution pattern of a double bond. Acting as a pair of chemical scissors, the reactive gas cuts the double bond and replaces it with oxygen atoms, i. e., carbonyl groups. However, ozonolysis does not afford information on the stereochemistry of the alkene if such stereochemistry existed originally. Thus, the generic, stereoisomeric alkenes 1 and 2 give rise to the same pair of carbonyl compounds. The astute reader will recognize that neither of these reactions is a balanced equation. Only two oxygen atoms (one equivalent of O2) is required to balance this reaction. The fate of the third oxygen atom will be considered later.
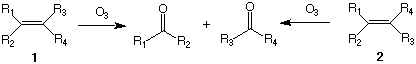
Ozonolysis is also an important reaction from the synthetic perspective with compounds that have several functional groups. Because alkenes are nucleophilic and carbonyl groups are electrophilic, aldehydes and ketones can be stored as alkenes during synthetic reactions that are electrophilic. At the appropriate time, the alkene can be converted to the aldehyde or ketone when it is its turn to undergo electrophilic reactions.
Acyclic Alkene Substitution Pattern:
The substitution pattern of a double bond in an acyclic alkene can be ascertained by the number and type of carbonyl compounds that are formed (see Table below). If the two carbons of the double bond contain the same groups, then only one carbonyl compound is formed. Thus, (E)- and (Z)-2-butene give only acetaldehyde. If the two carbons of the double bond are substituted differently, then two carbonyl compounds are obtained. --> Index
|
Double Bond Substitution |
Formaldehyde (CH2O) |
Aldehydes (RCHO) |
Ketones (R2CO) |
|
Unsubstituted (ethylene) |
2 |
- |
- |
|
Monosubstituted |
1 |
1 |
- |
|
1,1-Disubstituted |
1 |
- |
1 |
|
1,2-Disubstituted |
- |
2 |
- |
|
Trisubstituted |
- |
1 |
1 |
|
Tetrasubstituted |
- |
- |
2 |

Ozone reacts with an alkene via a cycloaddition reaction to form an unstable 5-membered ring called a molozonide. In the mechanism shown below, two C-O bonds are formed and the π-bond of the alkene is broken. The process is a syn addition of the electrophile ozone to the nucleophilic alkene. The electrons involved in the process are in blue. --> Index

The 5-membered ring of the unstable molozonide 4 has a plane of symmetry that passes through the central oxygen and the C-C bond. In the decomposition of the molozonide, not only is a weak O-O bond broken but a relatively strong C-C σ-bond is also broken. Because of the symmetry, there are two mechanistic pathways by which the molozonide can collapse. They are designated as the red and green mechanisms shown below. In each pathway one of the carbonyl compounds (5 and 8) is formed along with a species known as a zwitterion (6 and 7).

There are two interesting points about the zwitterion. First, it may be viewed as an oxidized form of an aldehyde or a ketone. [Do you remember the extra oxygen in the beginning of the discussion? More later.]. Secondly, a zwitterion is also a resonance structure [see below]. In fact, it is isoelectronic with ozone itself. One of the terminal oxygens of ozone has been replaced with R-C-R to form the zwitterion. --> Index

You may now ask, "If the zwitterions is isoelectronic with ozone, shouldn't it react similarly?" It does! When the ozonolysis is conducted in a non-nucleophilic solvent such as CH2Cl2, the zwitterions 6 and 7 undergo cycloaddition reactions with the carbonyl compounds 5 and 8 to form ozonides 9, 10 and 11, each of which can exist as a pair of diastereomers (cis/trans). Thus, ozonide 9 can be formed from carbonyl 5 and zwitterion 6 and from zwitterion 7 and carbonyl 8. In addition, ozonide 10 arises from carbonyl 5 and zwitterion 7 while ozonide 11 forms from carbonyl 8 and zwitterion 6. In fact, each of the ozonides can form two stereoisomers as long as th R-groups are different. Six stereoisomers does not present a problem because the ozonides will be decomposed in a subsequent reaction. In the mechanism below, the red and green bonds and unshared electron pairs are the ones involved in ozonide formation. --> Index

The conversion of an alkene to an ozonide is a 6-electron oxidation yet the oxidation of an alkene to two carbonyl compounds is a 4-electron oxidation. A 2-electron reduction of the ozonides is required. [See Oxidation Levels]. In other words, one of the oxygens of the ozonide must be reduced. The O-O bond of an ozonide has one of the oxygens more electron deficient than the other. The two electron reduction of the O-O bond of the ozonide is often accomplished with zinc or dimethyl sulfide. The mechanism below illustrates how the ozonide is reduced by two electrons while the dimethyl sulfde is oxidized by two electrons to dimethyl sulfoxide 13. --> Index

Cyclic Alkene Substitution Pattern: (added 10/24/01)
The carbonyl compounds obtained from cyclic alkenes are the
same as that obtained from acyclic alkenes except that both
carbonyl groups are in the same molecule. That is, they are
tethered together by the carbon chain that made the ring having an
endocyclic (within the ring) double bond. Several examples
follow:

|
The 1,2-disubstituted double bond of cyclohexene 14 must give two aldehyde groups. That they are in the same molecule and separated by four methylene groups tells us that the dialdehyde 15 came from a ring wherein the double bond was endocyclic. Normally, we would expect the product to be a 6-membered ring. |
|
Unlike acyclic 1,2-disubstituted alkenes where we cannot determine whether or not the original double bond was (E)- or (Z)-, in this instance we can exclude the possibility of an (E)- double bond in cyclohexene because of ring strain considerations. If the ring has seven atoms or fewer, (E)- double bonds are excluded. There is, however, a caveat. In the absence of any information other than that derived from the ozonolysis, cyclododecadiene 16 and its higher homologs satisfy dialdehyde 15. (E)- and/or (Z)- double bonds are possible.
The trisubstituted double bond of the cyclopentene 17 leads to an aldehyde and a keto group in the same molecule, namely, ketoaldehyde 18. Clearly, the three methylene groups dictate a 5-membered ring.

In the case of diene 19, the double bonds are exocyclic
to the ring. This means that they behave as two acyclic double
bonds giving "two" separate carbonyl compounds each. But two of
the carbonyl groups are in the diketone 20, giving three
compounds instead of four. To determine the structure of the
compound(s) from which 20 and 21 derive upon
ozonolysis, we work backwards. The two acetaldehydes 21
cannot be reassembled to form either of the two 2-butenes while
diketone 20 is reconstituted to the bicyclohexene 22
(strain energy = ~a gazillion kcal/mol) because the mental
exercise would give two compounds, not one. Clearly, the solution
must be diene 19, or its stereoisomer 23. By the
way, if 19 and 23 are stereoisomers, then there must
be a difference in the double bond, right?



