Fig. 1 |
Fig. 2 |
|---|
Adamantane: Adamantane (C10H16), a tricyclic alkane with a diamondoid structure, was first isolated in 1933 in minute amounts from petroleum [Landa, Macháček]. The first chemical synthesis of adamantane was inefficient [Prelog, Seiwerth;, 1941]. In 1957 Schleyer reported an efficient synthesis of adamantane by Lewis acid-catalyzed rearrangement of the tetrahydro derivative of the cyclopentadiene dimer. Adamantane (Fig. 1) is comprised of three chair cyclohexane rings. [Although you may see four, formation of three rings consumes all of the carbon atoms. The "fourth ring" is an artifact. Recall that adamantane is tricyclic (Degree of Unsaturation = 3). Click on "show carbon #". For example, breaking the C1-C2 bond forms a bicyclic species. Subsequent cleavage of the C7-C10 bond affords a monocycle. Lastly, fragmentation of the C6-C7 bond provides an acyclic structure.] The carbon atoms are of two types. The six red carbons (C2, C4, C6, C8, C9 and C10) are methylene groups while the blue carbons (C1, C3, C5 and C7) are methine carbons. Adamantane is achiral having severalmirror planes and axes of rotation. Rotate the structure so that you are viewing down the C2-C6 axis. Introduction of a substituent at both of these remote methylene atoms will create a chiral molecule. [Owing to symmetry, the pairs C4-C8 or C9-C10 could have utilized.] Adamantane-2,6-diol: Fig. 2 represents one enantiomer of adamantane-2,6-diol. Click "show carbon #" and "zoom in" in Fig. 2 and align the structure so that you are viewing down the C2-C6 axis with the C2 hydroxyl pointing to "noon". The hydroxyl at C6 will point to three o'clock. The same result will occur looking down the C6-C2 axis. This structure is not superimposable on its mirror, which can be formed by inverting the stereochemistry at either C2 or C6. Both C2 and C6 are stereogenic and chirotopic atoms. Click on "show R/S". Carbons 2 and 6 are both of the S-configuration and equivalent to one another. Rotate C2 to the position occupied by C6 and then roll the structure from top to bottom by 90o. With a little tweeking you have duplicated the original structure.
|
|---|
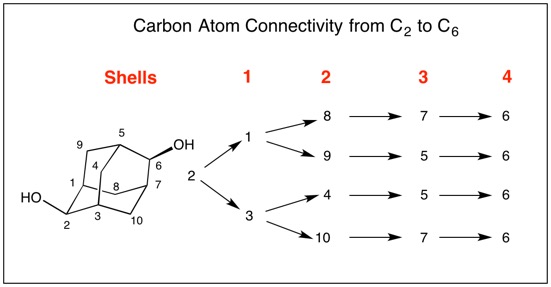
A Brief Interlude: Return Fig. 2 to the "noon- three o'clock position". Now look down the C1-C3 axis (Fig. 3) of chiral 1,3-dimethyl-1,2-propadiene (symmetrical dimethylallene) placing the methyl groups in the "noon- three o'clock position". Since the hydroxyl groups in the adamantane and the methyl groups in the allene occupy the same spatial arrangement, one might presume that they bear the same descriptors. such is not the case. In adamantane-2,6-diol it is the chirotopicity of C2 and C6 that is at issue. There is no chiral atom in the dimethylallene but rather molecular chirality, M vs. P applies here (click here for chirality of allenes and biphenyls). [Note: The P chirality in Fig. 3 is occasionally designated as aR (axial R) meaning that the methyl groups form a right handed helix. The CIP rules originally defined this chirality as S. The priorities of the substituent atoms are Cnear> Hnear> Cfar> Hfar or, as you look down the C1-C3 axis, C4 > H6 > C5 > H7 by hovering your cursor over the atoms.] Use of Ro and So Designations: So how are C2 and C6 assigned the S-configuration? There are four pathways via the carbon framework that lead from C2 to C6 are illustrated in Fig. 4.
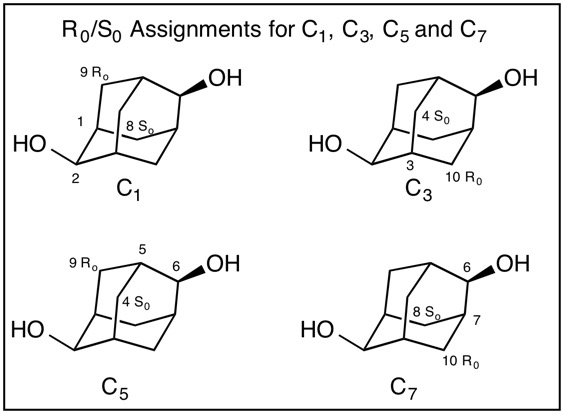
Fig. 4 Clearly, C2 bears a hydrogen (lowest priority) and an oxygen (highest priority) atom (CIP rule 1). But which two carbon pathways constitute the second and third priorities? CIP rules 4 coupled with the assignment of temporary R/S (Ro/So) configurations are utilized to resolve the apparent impass. First, temporary configurations are assigned at C6 (Fig. 5). Applying the right hand rule assigns C7=Ro and the left hand rule makes C5=So. [For a review of the right and left hand rules, click here.] The configuration of C5 (Fig. 5) has the hydrogen as the lowest priority atom and C6 is the highest. Consequently, C9=Ro and C4=So while C7 assigns C10=Ro and C8=So. Structure C5,6,7 displays the temporary assignments. Fig. 6 expands Fig. 4 by incorporating the temporary assignments Ro/So. Application of CIP rule 4 eliminates RoSo and SoRo because they are unlike (ul) matches leaving RoRo and SoSo (lk). Both of these assignments are common to C3 while the unlike pair are associated with C1. Therefore, the configuration at C2 follows the order O > C3 > C1 > H. C2 is of the S-configuration! Owing to the symmetry in adamantane-2,6-diol, C6 is also of the S-configuration. Configurations of Methine Carbons; C1, C3, C5, C7: In the evaluation of the configuration of C1 (Fig. 7) the hydrogen has the lowest priority and C2 has the highest priority. The right hand rule assigns C9=Ro while the left hand rule leads to C8=So. Therefore, C1 is of the R-configuration (C2 > C9 > C8 > H. Similar analysis of C3, C5 and C7 demonstrates that these carbons are also of the R-configuration. The configuration of adamantane-2,6-diol is 1R,2S,3R,5R,6S,7R. [Note: ChemBioDraw 13.0.2.3020 employs a different algorithm leading to (1R,2S,3S,5R,6S,7S)-adamantane-2,6-diol for this enantiomer.] |
Fig. 3 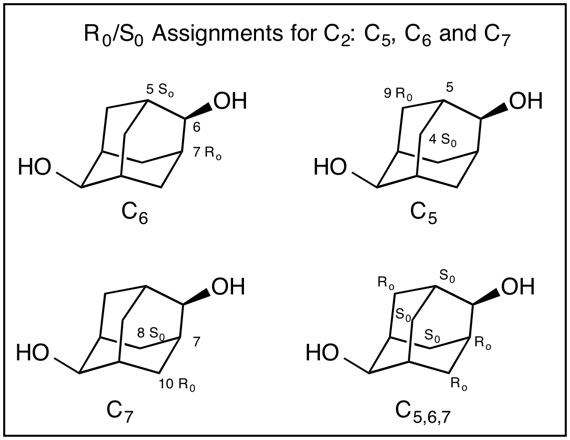 Fig. 5
Fig. 7 |
|---|