|
Fig. 1
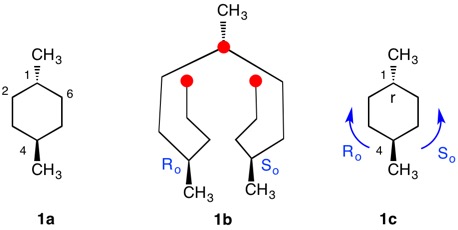
trans-1,4--Dimethylcyclohexane |
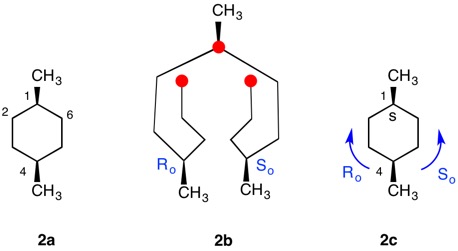
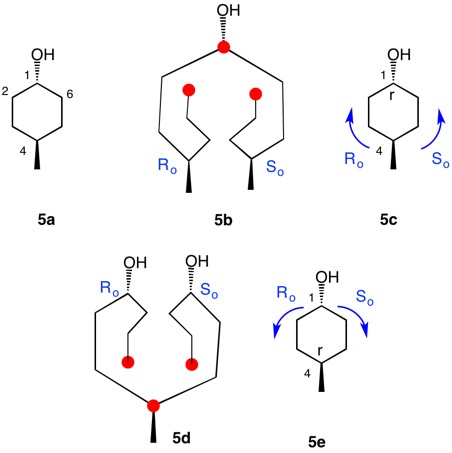
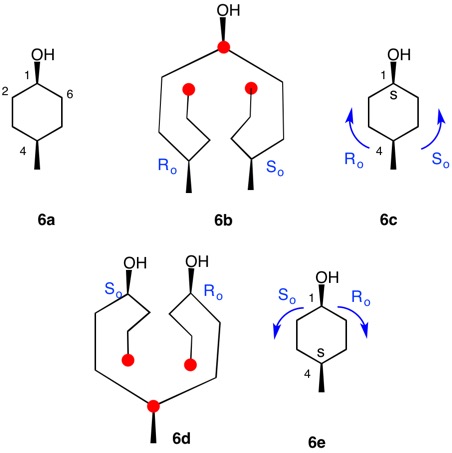
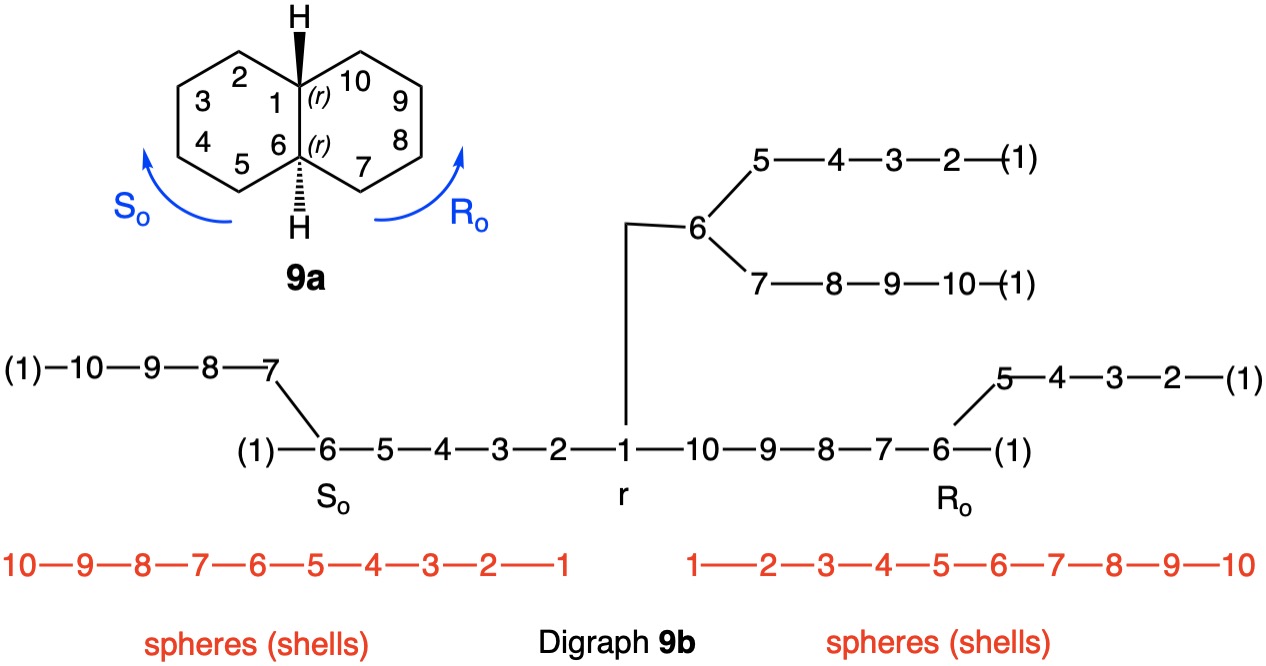
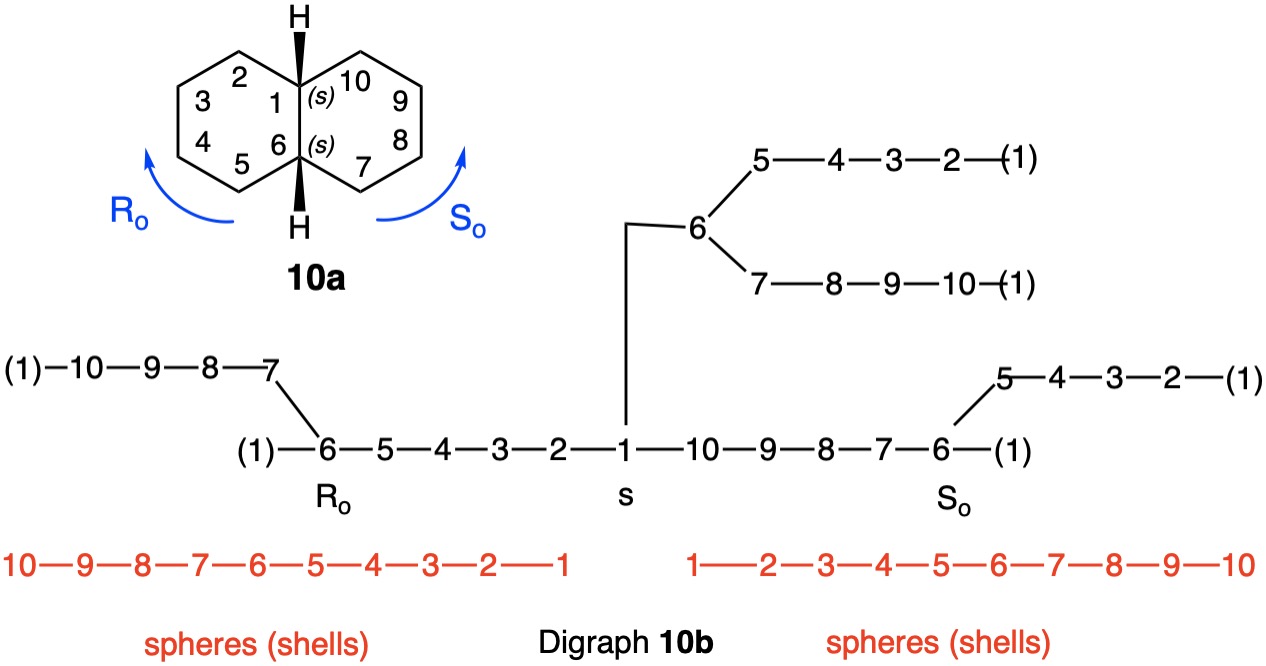
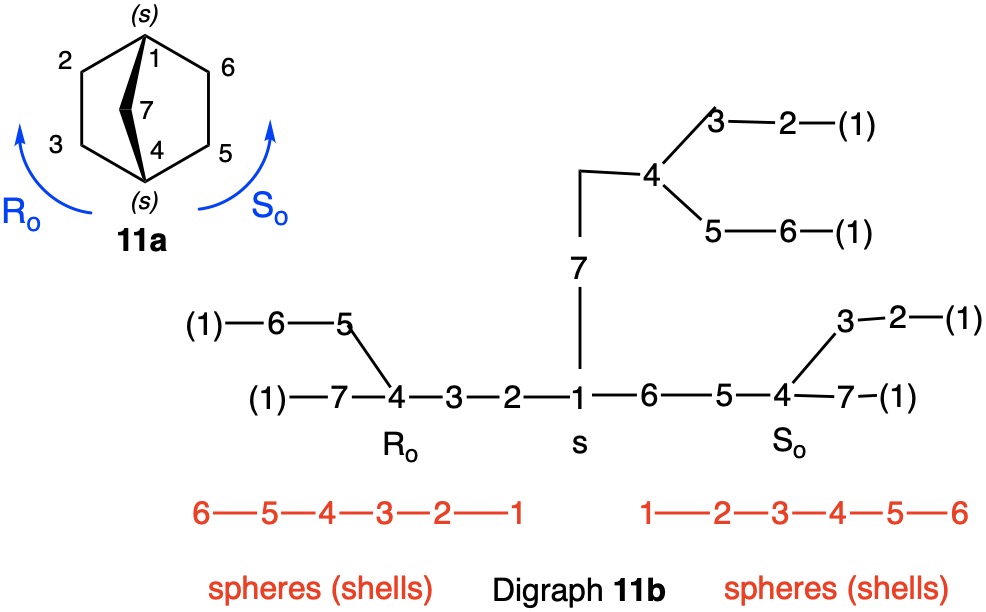
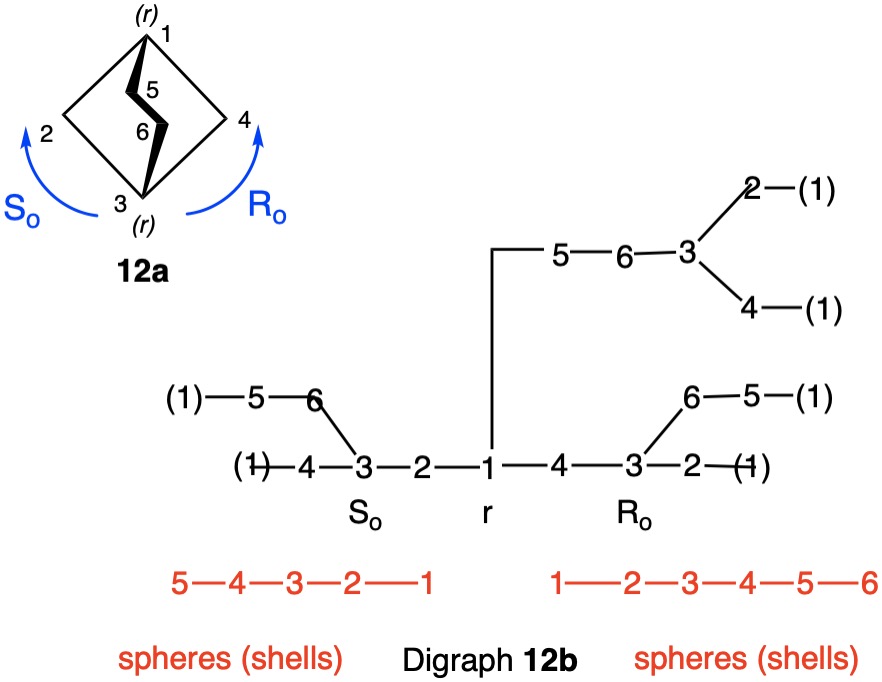
The trans-stereoisomer (Fig. 1), as represented in 1a, has a plane of symmetry through C1 and C4 and a two-fold axis of symmetry along the x-axis that passes through the center of the C2-C3 and C5-C6 bonds. The pseudo-asymmetric carbons C1 and C4 are equivalent owing to the two-fold axis. The configurational assignments will be the same. At C1 the lowest priority (4) atom is hydrogen and the methyl group ranks third. But how does one chose between the chains C2-C3-C4 and C6-C5-C4 for the remaining two priorities? They are essentially mirror images of one another. Therein lies a solution! Digraph 1b is constructed by following the path from C1 (red dot) to C2-C6 and terminating in a duplicate carbon atom (C1; red dot) that has an atomic number of six but is attached to three "phantom" atoms of atomic number zero. The same process may also proceed from C1 to C6-C2 terminating in a duplicate C1. These two chains are enantiomeric and temporary configurations (Ro/So) can be assigned to each based on the "chiral" center at C4. The lefthand C4 of 1b is assigned an Ro configuration with priorities C3-C2-C1>C5-C6-C1(duplicate)>CH3>H. Conversely, the enantiomeric righthand C4 chain is of the So configuration. Therefore, C1 has the top two priority chains as Ro and So. Because R has priority over S, the same is true for Ro and So. The configuration of C1 is r given Ro>So>CH3>H. By symmetry C4 is also of the r-configuration. Structure 1c is a condensed version of 1b. cis-1,4-Dimethylcyclohexane (Fig. 2) has a plane of symmetry through C1 and C4 and a two-fold axis of rotation orthogonal to the plane of the ring. Both of these pseudoasymmetric centers will have the same configuration. The only difference between digraph 2b and 1b is the stereochemistry at C1 while the Ro/So assignments remain the same. The priorities remain the same, Ro>So>CH3>H. Both C1 and C4 have the same s-configuration. |
Fig. 2
cis-1,4-Dimethylcyclohexane |
|---|