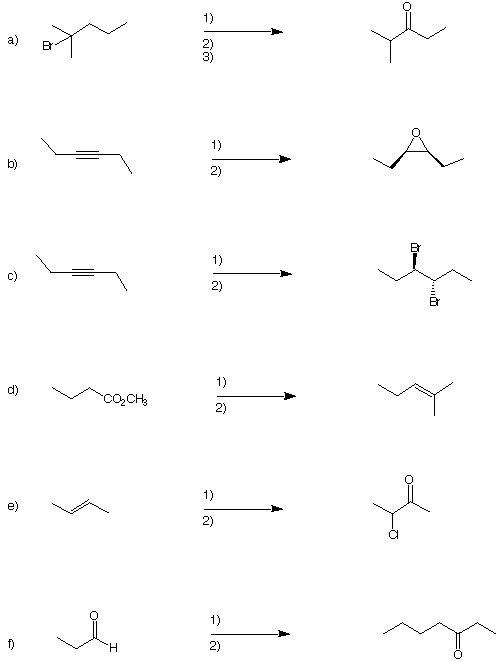FINAL EXAMINATION
CHEMISTRY 220a
Thursday, December 16, 1999
9:00 A.M.
NAME (print):
______________________________________________________________
TA:_____________________ Section Day:_________________ Section
Time:__________
Take a few moments to look over the exam. Do problems first with
which you are most
comfortable. Important points and unknowns are in bold type. Do
all preliminary work on
the worksheets. The worksheets will not be graded. There is a
Periodic Table on page 16 of the
exam. The exam is 2 to 2-1/2 hours with 1/2 hour for review. STOP
writing when you are
asked to do so. Put your name on the cover sheet and subsequent
pages where indicated.
Bonus: What is the name of the compound whose structure is on the
front of your textbook?
You are to do either 1a or 1b, not both of them.
REMEMBER: Neatness is to your advantage. Have a GREAT winter
break! Best of
Luck in Chem. 221b!
|
1. (35 pts) Mechanism __________
2. (25 pts ) Reactions I __________
3. (25 pts) Reactions II __________
4. (32 pts) Potpourri __________
5. (30 pts) Radicals __________
6. (30 pts) NMR __________
7. (30 pts) Structure Proof __________
8. (43 pts) Synthesis/NMR __________
9. (5 pts) Bonus __________
___________________________________
Total
|
Let's hear it for cyclohexane!!!
|
1a) Mechanism (35 pts.): You are to do either 1a or 1b, not both.
Mark the box in
the left margin for 1a to be graded.

Nature employs the SN2 reaction twice in the formation of -OCH3
and -NCH3
groups where oxygen and nitrogen, respectively, are nucleophiles.
Adenosine triphosphate
(ATP) 1 reacts with the amino acid methionine 2 to form S-adenosyl
methionine (SAM) 3,
Nature's methylating agent. For instance, SAM serves as a
methylating agent to convert
dihydroxy aldehyde 4, acting as a nucleophile, into vanillin
5.
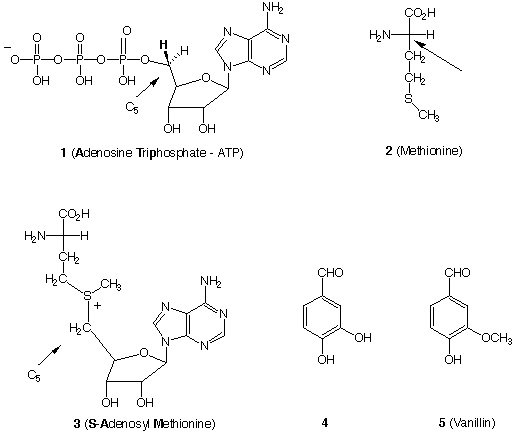
a) (5 pts.)Why is the triphosphate group (or a phosphate group for
that matter) in 1 a
good leaving group?
b) (5 pts.)What is the configuration (R,S) at the carbon
designated by the arrow in
methionine 2 (Fischer projection)? ______
The mechanism of this reaction sequence has been unraveled using
isotopic labeling.
Assume that the bold hydrogen at C5 in ATP 1 is a deuterium atom
instead of hydrogen.
Answer the following questions.
c) (5 pts.)What is the configuration (R,S) at C5 in deuterium
labeled ATP 1 ? _____
d) (5 pts.)What is the configuration (R,S) at C5 in deuterium
labeled SAM 3? _____
Assume that labeled methionine 2 is prepared such that the -SCH3
group is now a chiral
methyl group -SC(HDT) of the R-configuration containing hydrogen,
deuterium and
tritium.
e) (5 pts.) Draw this configuration using the template below.
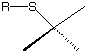
f) (5 pts.) What is this configuration in the labeled methyl group
in SAM 3? ______
g) (5 pts.) Draw the configuration of the labeled methyl group in
vanillin 5 using the
template shown below. What is its configuration? ________
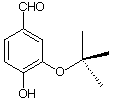
1b) Mechanism (35 pts): You are to do either 1a or 1b, not both.
Mark the
box in the left margin for 1b to be graded.

During the term we studied the oxidation of alcohols with Cr(VI)
reagents. In
particular, the use of PCC to oxidize primary alcohols to
aldehydes was addressed.
The sequence of reactions in equations a - c is a variation of the
so-called Swern
oxidation, which accomplishes the same goal. Your counterparts at
"that other
institution" were asked questions about this series of reactions
during the past term.
The questions in italics were asked of them.
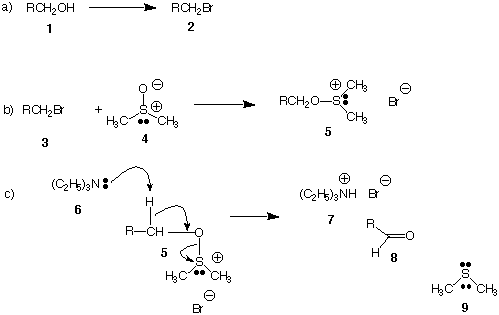
a) (5 pts.) Provide an alternative to HBr for the conversion of
alcohol 1 to
bromide 2 in equation (a). [We have discussed two alternative
methods]
b) (5 pts.) Suggest a mechanism for the reaction of dimethyl
sulfoxide 4 to form
sulfoxonium salt 5 in equation (b). Use the curved arrow
formalism. [The
polarized S-O bond in 4 is the same as S=O.
c) (5 pts.) Classify the reaction in equation (b): (SN1,
SN2, E2, E1, addition,
etc.)
d) (5 pts.) What is the oxidizing agent? What is the
reduction product?
They were asked to provide a mechanism for the decomposition of
the
sulfoxonium salt 5 formed in equation (b). They were given as the
correct answer
(after the exam) the E2 mechanism shown in equation (c). However,
the
mechanism shown below (equation d) is equally plausible. In this
mechanism,
intermediate 10 is formed. The same reactants 5 and 6 give the
same products 7,
8, and 9 in both mechanisms! To decide between equation (c) or
equation (d) as
the correct mechanism, dimethyl sulfoxide 4 [(CD3)2S=O]
having deuterium (six
of them) in place of hydrogen was used.
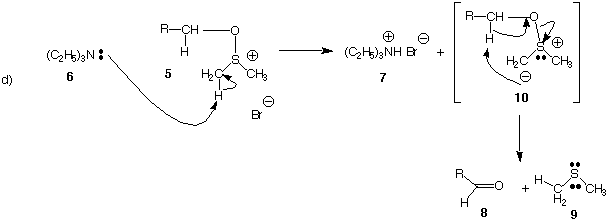
• Here are the facts!
When totally deuterated dimethyl sulfoxide 4 [(CD3)2S=O]
is used to form
sulfonium salt 5, its reaction (i.e., 5) with triethylamine 6
gives ammonium salt 7
[(C2H5)3ND+ Br-] containing one deuterium atom and
dimethyl sulfide 9
[CD2HSCD3] containing only five of the original six
deuterium atoms that were
present in the (CD3)2S=O.
e) (15 pts.) Which of the two mechanisms does the data support?
[Hint:
Compare the location of the deuterium atoms in equation (c) and
(d).]
Illustrate and explain.
2) Reactions I (25 pts.) Provide the expected major product in
five of the following six
reactions. Do only five. If you did six, put a big "X" through the
one you want
excluded. Pay attention to stereochemistry.
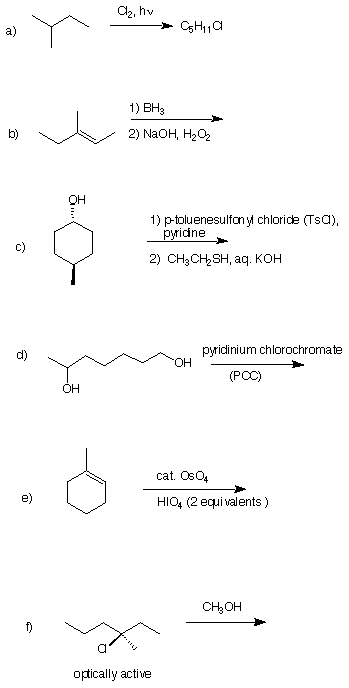
3) Reactions II (25 pts.): Provide the reagents in five of the
following six reactions. Do
only five. If you did six, put a big "X" through the one you want
excluded. Pay
attention to stereochemistry. Each reaction requires the number of
steps shown.
(There may be alternatives.) Place the reagents by the arrows.
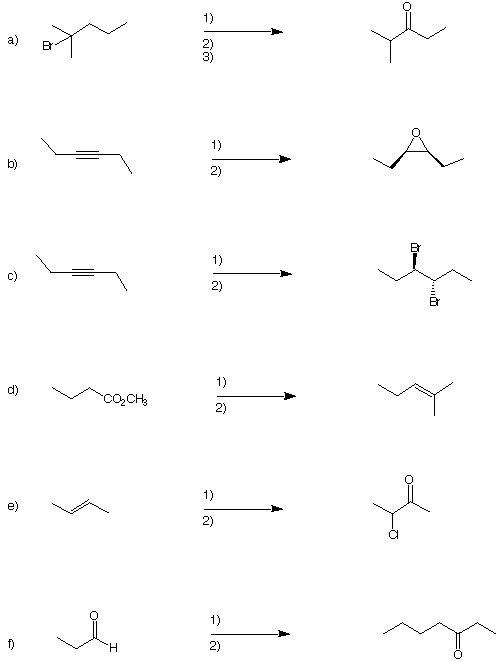
4) Potpourri (32 pts.): Circle the correct answer in each of the
following:
a) The compound whose conjugate base will deprotonate only two of
the other
compounds. [Hydrogens are in bold type.]
C2H2 NH3 (CH3)2CHOH NH4Cl CH3CO2H
b) The compound with the shortest bond (bold):

c) The best estimate of the rotational barrier (kcal/mol) about
the C-C bond in ethane.
0.9 ---------3.0 ----------3.3
----------------3.9------------------- 5.0
d) The hybridization of the central carbon of allene
(CH2=C=CH2).
s ----------p------------- sp----------------
sp2----------------sp3------------------ p
e) The heat of formation of n-butane is -30 kcal/mol. The heat of
hydrogenation (2 moles)
of 1-butyne is -70 kcal/mol. The best estimate for the heat of
formation (kcal/mol) of 1-
butyne is:
-100 ..............70 ...............-45....................
-40............... +39.............. +30
f) A one hydrogen triplet appears in a 1H NMR spectrum with J =
6.0 Hz. The three
peaks appear at d 0.9, 1.0, and 1.1. What is the frequency (MHz)
of the spectrometer?
indeterminate .............500 ..............100...............
600.................. 60
g) The value of DGo (kcal/mol) for the equilibrium between the two
chair conformations
of trans-1,4-dimethylcyclohexane (gauche butane interaction: 0.9
kcal/mol).
0------------0.9------------- 1.8---------------- 3.6
------------5.0 ---------------none of these
h) The compound with the greatest number of singlets in its broad
band proton
decoupled 13C NMR spectrum.
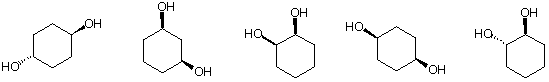
5) Radicals (30 pts.): The free radical chain chlorination of
cyclohexane affords
chlorocyclohexane. Answer the following questions.
a) (10 pts) Write the propagation steps?
propagation step 1 --->
propagation step 2 --->
b) (10 pts) Determine the CH bond dissociation energy (BDE) for
cyclohexane, the
BDE for the CCl bond in chlorocyclohexane, and the DHo
for propagation step 2.
Show work. [DHo (overall rxn.) =
-30 kcal/mol; DHo (propagation step 1) =
-8
kcal/mol; BDE HCl = 103 kcal/mol; BDE Cl2 = 58 kcal/mol]
d) (10 pts) Provide an energy diagram for this reaction. Include
the three values of
DHo and obey the Hammond postulate.
6) NMR (30 pts.): An unknown compound A (C8H16) reacts with O3
followed by zinc
reduction to give two compounds, B and C. Compound B has a singlet
(d 2.07) in its 1H
NMR spectrum and two singlets (one singlet is weak; d 204) in its
broad band proton
decoupled 13C NMR spectrum. The 1H NMR spectrum of C displays the
following data
(d values are estimated): d 2.5 (quartet) and 1.2 (triplet). The
integration of the two signals
in the spectrum of C is in a ratio of 2:3 (quartet:triplet). What
are the structures of A, B,
and C? Explain.
7) Structure Proof (30 pts.): (S)-(+)-Limonene, (C10H16,
[a]D +124o) is a terpene found in
the rind of lemons and oranges. Ozonolysis of (S)-(+)-limonene
followed by treatment
with dimethyl sulfide results in the isolation of 1. Hydrogenation
of limonene gives cis-
and trans-1-isopropyl-4-methylcyclohexane.
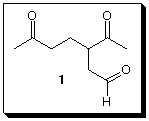
a) ( 5pts.) How many degrees of unsaturation are present in
limonene?
b) ( 5pts.) What important information is gained from the
hydrogenation experiment?
c) (5 pts.) What is the other product of ozonolysis?
d) (5 pts.) Draw (S)-(+)-limonene.
e) (10 pts.) (S)-(+)-Limonene reacts with 2 equivalents
of HBr in Markovnikov
fashion to give two optically inactive compounds C10H18Br2. Draw
their
structures and explain briefly. {Hint: Think about the
hydrogenation
experiment.]
8) Synthesis (43 pts): Gainfully employed in a laboratory during
the summer, a graduate of
Chem 220a is given the challenge of synthesizing disparlure (1),
the optically-active sex
attractant of the female gypsy moth. She uses 5-methyl-1-hexanol
(3), part of oleic acid 2
for the n-alkane chain, and compounds with two or fewer carbon
atoms as her sources of
carbon. All other reagents are available to her.
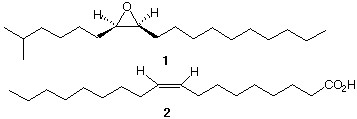
a) (25 pts) Show her synthetic plan.
b) (10 pts.) She understands why her starting alcohol 3 (C7H16O)
contains only six
singlets in its broad band proton decoupled 13C NMR spectrum, and
she fully expects to
see all nineteen carbons as unique singlets in the 13C spectrum of
her synthetic disparlure
1. [Note: There is no fortuitous overlap of singlets].
Explain her reasoning.
c) (8 pts.) The 1 H and 13C NMR spectra of her synthetic material
were identical to the
corresponding spectra of the pheromone isolated from the female
gypsy moth. What
measurement that she took showed a difference between the
synthetic and natural material?
Why?
9) Bonus (5 pts) What is the name of the compound whose structure
is on the front of your
textbook?