NAME (print): ___________________________________________________
TA:_____________________ Day:_________________ Section Time:__________
Important points are in bold.
Complete the section above and put your name on pages 2-6.
Take a few moments to look over the exam. Answer each question on the exam paper.
Do all preliminary drawing or computations on the Work Sheets at the end of the exam.
They will not be graded.
A Periodic Table is on page 7 of the exam should you need it.
The exam is 55 minutes.
STOP writing when you are told to do so.
REMEMBER: Neatness is to your advantage.
1. (25 pts) ______
2. (20 pts) ______
3. (20 pts) ______
4. (20 pts) ______
5. (15 pts) ______
________________________________
Total (100 pts)
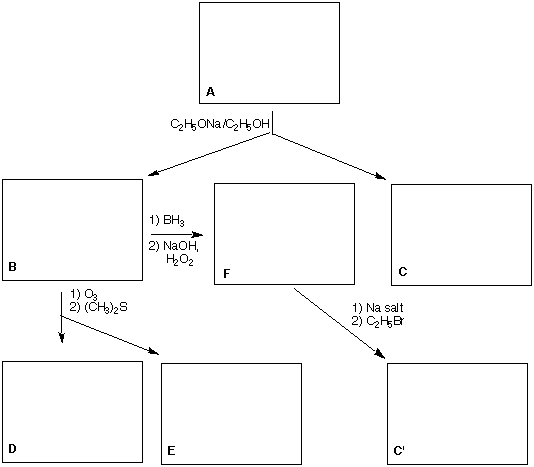
1) (25 pts) An optically-active compound (R)-A (C5H11Br) reacts with C2H5ONa in C2H5OH to give B
(C5H10) and C (C7H16O). Ozonolysis and dimethyl sulfide reduction of B produces ketone D and
aldehyde E. Ketone D is shown not to be acetone (2-propanone)! Hydroboration and alkaline hydrogen
peroxide oxidation of B gives F (C5H12O). The sodium salt of F reacts with ethyl bromide to form C',
which differs from C in one way.
a) Place the structures of A-F in the appropriate boxes with any brief commentary.

b) How do C and C' differ?
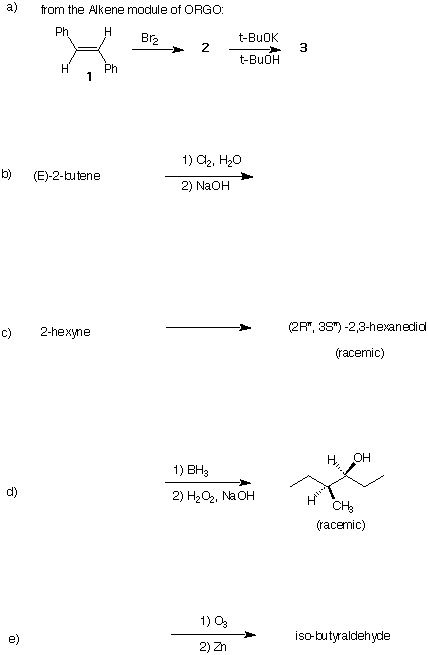
2) (20 pts) Design an efficient synthesis of 2-hexanone (a ketone) from acetylene and ethylene. All
reagents are available to you.
3) (5 x 4 pts = 20 pts) In each of the following reactions, provide the reactant, product, or reagents as
required. If there is more than one possible product or reactant, include it. Pay attention to
stereochemistry.
4) (5 x 4 pts = 20 pts) In each of the following questions, circle the appropriate answers.
a) The best estimate (kcal/mol) for the DHfo of 2-butyne given that its DHo of hydrogenation to n-
butane is exothermic by 65 kcal/mol. [DHfo = -30 kcal/mol for n-butane]
+65 -95 -35 +35 +5 -5
b) Sodium acetylide will deprotonate which of the following acids
acetylene ethylene ammonia ethanol ethane ammonium ion
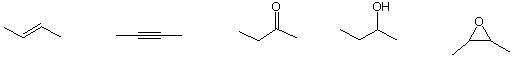
c) The alkene that liberates the greatest amount of heat upon hydrogenation
![]()
d) The reagents that give stereospecific reactions with both 2-butenes
CHCl3/NaOH BH3;NaOOH Br2/CCl4
OsO4/H2O2 Zn(Cu)/CH2I2
e) The most highly oxidized structures
|
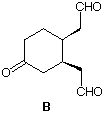
5) (15 pts) Sunday night is turning into Monday morning as two Chem 220a students work on the last problem of their Problem Set. Compound A (C10H14O) undergoes ozonolysis to give the cis-ketodialdehyde B. He claims that because there are three carbonyl groups and no plane of symmetry in B, there are three possible structures for A. She says that his reasoning is conceptually correct but there is only one structure that can represent compound A. Outline their respective arguments.
|
|