1. (26 pts) ______
2. (24 pts) ______
3. (15 pts) ______
4. (20 pts) ______
5. (15 pts) ______
________________________________
Total (100 pts)
 The
Tetrahedral Carbon
The
Tetrahedral Carbon
NAME (print): ___________________________________________________
TA:_____________________ Day:_________________ Section Time:__________
Take a few moments to look over the exam. Answer each question on the exam paper.
Important points are in bold.
Do all preliminary drawing or computations on the Work Sheets at the end of the exam.
They will not be graded.
A Periodic Table is at the end of the exam should you need it.
The exam is 55 minutes.
STOP writing when you are told to do so.
REMEMBER: Neatness is to your advantage.
|
1. (26 pts) ______
2. (24 pts) ______
3. (15 pts) ______
4. (20 pts) ______
5. (15 pts) ______
________________________________
Total (100 pts)
|
 The
Tetrahedral Carbon The
Tetrahedral Carbon
|
1) (26 pts) The terpene limonene (C10H16) 1, a constituent of lemon grass, reacts with 2 equivalents of
hydrogen in the presence of a catalyst to produce two compounds, 2 and 3, both of which have the
molecular formula C10H20.
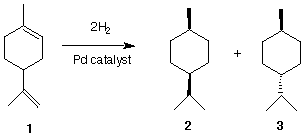
a) (2 pts) In a single word, what is the relationship between cycloalkanes 2 and 3?
b) (12 pts) Draw a 3D view of each cycloalkane in its two chair conformations with the more stable and
less stable conformations in their proper location. [Note the direction of the equilibrium arrows.]
2 --->
![]()
3 --->
![]()
Problem 1 is continued on the next page
c) (12 pts, equally weighted) One of the cycloalkanes has an energy difference (DGo) of 0.4 kcal/mol
between its chair conformations. Which cycloalkane is it?
i) Structure number goes here ---> __________ Is it cis or trans? __________
Given DGo=1.7 kcal/mol for the difference in energy between the axial and equatorial chair conformations
of methylcyclohexane, what is the value of DGo for isopropylcyclohexane? Show work.
ii) Answer goes here -----> __________ kcal/mol
What is the difference in energy (DGo) between the chair conformations of the other cycloalkane (not the
one in 1ci above)? Show work.
iii) Answer goes here ---> __________ kcal/mol
2) (24 pts) To analyze the barrier of rotation about the C2-C3 bond of 2,3-dimethylbutane, there are three
staggered conformations and three eclipsed conformations to consider. [Data: H/H eclipsed, 1.0 kcal/mol;
CH3/H eclipsed, 1.3 kcal/mol; CH3/CH3, eclipsed, 3.0 kcal/mol; CH3/CH3, gauche, 0.9 kcal/mol]
a) (12 pts) Draw a Newman projection of the most stable conformation and determine its energy.
Label interactions with their appropriate values.
b) (12 pts) Draw a 3D (sawhorse) structure of the least stable conformation and determine its
energy. Label interactions with their appropriate values. [You may use "CH3" as opposed to drawing
the methyl groups. [You need only draw the bonds with their substituents emanating from C2 and C3.]
3) (15 pts) Circle the best answer(s) or follow specific instructions.
a) C-H bond length in an alkane
1.54 Å 154o 1.1 cm 1.1 Å 109o 28'
b) Compounds with a net dipole moment
![]()
c) Number the following acids in order of increasing pKa value (lowest = 1, highest = 5).
HBr ammonia acetic acid cyclopentane methanol
d) This year, 1999, is the 125th anniversary of the tetrahedral carbon concept proposed by
Kekule van't Hoff Liebig Baeyer Berzelius
e) Which of the following term(s) applies to cyclopropane?
CH bond eclipsing flagpole interaction angle strain
gauche interactions resonance
4) (20 pts) Structure 1 is a poor rendering of ethylene (ethene).
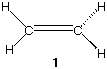
a) (5 pts) What is wrong with structure 1 as it is drawn?
b) (15 pts) Use molecular orbitals to provide a proper representation of ethylene. Include and label
orbitals, bond angles (approximate) and types of bonds.
5) (15 pts, equal weight) Complete the following: