NAME (print): ____________________________________________________________
TA:_____________________ Section Day:_______________ Section Time:__________
Take a few moments to look over the exam. Answer each question on the exam paper. Do the questions with which you are most comfortable first.
The exam is 100 points . Important items are in bold.
Do all preliminary drawing and computations on the work sheets at the end of the exam. The work sheets will not be graded. If you run out of space, write on the back of the preceding page.
The exam is 50 minutes. STOP writing and hand in your exam when you are asked to do so.
Remember: Neatness is to your advantage.
1. (20 pts) ______
2. (20 pts) ______
3. (20 pts) ______
4. (20 pts) ______
5. (20 pts) ______
______________________________________
Total (100 pts)
1. (4 x 5 pts. = 20 pts.) Provide the correct answer for each of the following questions.
a) The four signals of a quartet are located at d 3.25, 3.27, 3.29 and 3.31 in a 1H NMR spectrum taken at 300 MHz. What is the value of J? Show work.
b) A coupling constant has a value of 10 Hz at 300 MHz. What is the value of J at 60 MHz? Show work.
c) How many signals are displayed in the proton decoupled 13C NMR spectrum of 2,4-dimethylpentane? Illustrate.
d) A doublet of doublets appears in a 1H NMR spectrum. The spacing between the four peaks is 4 Hz, 8 Hz and 4 Hz (left to right). What are the values (in Hz) of the two coupling constants? Illustrate.
2. (20 pts.) The esters methyl propionate (C4H8O2) and ethyl acetate (C4H8O2) have the same number of unique chemical shifts and the same multiplicity of signals in their respective 1H NMR spectra. Draw the structure and the spectrum of each ester below with the correct order of chemical shifts and the correct multiplicity. Provide the area (in a circle above the signal) for each set of unique protons. The value of J in both compounds is the same. Estimate its value within one Hz.

The value of J -----------------------> ______________
3. (20 pts.) An unknown compound A (C7H14) reacts with ozone followed by reduction with dimethylsulfide to give compounds B and C. Compound B has the following 1H NMR spectrum: d 1.00 (triplet, J = 7 Hz) and 2.38 (quartet, J = 7 Hz). The integrated area of the triplet relative to the quartet is 3:2. The proton decoupled 13C spectrum of B displays 3 singlets, one of which is weak and located at ~200 ppm. What are the structures of A, B and C? Show your reasoning.
A -----> __________________
B -----> __________________
C -----> __________________
4. (5 x 4 pts. = 20 pts.) In each of the following problems a product, reactant or reagent is missing. Fill in the missing item(s). Be very careful about stereochemistry.
a) [See problem #2 under alcohols in ORGO.]
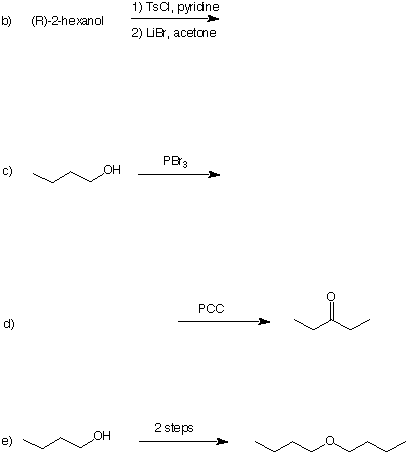
5. (20 pts.) Show how the ketone, 2-hexanone, can be prepared from ethanol and ethylene as the only carbon sources. All reagents are at your disposal.