NAME (print): ___________________________________________________
TA:_____________________ Day:_________________ Time:__________
Take a few moments to look over the exam. Answer each question on the exam paper.
The exam is 100 points with 12 optional bonus points. You can earn 112 points on a 100 point exam.
Important items are in bold.
Do all preliminary drawing or computations on the work sheets at the end of the exam..
The exam is 55 minutes.
STOP writing and hand in your exam when you are asked to do so.
Remember: Neatness is to your advantage.
1. (25 pts) ______
2. (25 pts) ______
3. (24 pts) ______
4. (10 pts) ______
5. (16 pts) ______
6. Bonus (12 pts) ______
_______________________________
Total (100 pts)
1. ( 25 pts) Consider trans-1-chloro-3-ethylcyclohexane.
a) (10 pts) Draw the chair conformations below so that the equilibrium arrows are correct. Be sure groups are clearly axial or equatorial.
chair A chair B
b) (7 pts) What is the value of DGo for the above equilibrium given the following DGo values for the equilibrium between axial and equatorial chair conformations of monosubstituted (X) cyclohexane: X = Cl = 0.5 kcal/mol; X = C2H5 = 1.9 kcal/mol? Show calculations.
c) (8 pts) Given that DGo = -1.4 log10 Keq, what percentage of each chair conformation is present? Show work.
Chair A = ___________
Chair B = ___________
2. ( 25 pts) Consider compound 1.
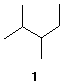
a) (5 pts) Name compound 1 using IUPAC rules.
b) (10 pts) Draw the two most stable conformations of this compound viewing from C2 to C3 (Newman projections). Conformer A is less stable than conformer B.
Conformer A Conformer B
c) ( 10 pts) Calculate the energies (kcal/mol) for each conformation in b) using the following data: H/H, eclipsed, 1.0 kcal/mol; CH3/H eclipsed, 1.3 kcal/mol; C2H5/H, eclipsed, 1.1 kcal/mol; CH3/CH3, eclipsed, 3.0 kcal/mol; CH3/CH3, gauche, 0.9 kcal/mol; CH3/C2H5, gauche, 1.0 kcal/mol. Place the appropriate energies in the Newman projections above. Place your answer below. Show calculations.
Conformer A _________
Conformer B __________
3) (24 pts) Circle the best answer(s) in each of the following problems.
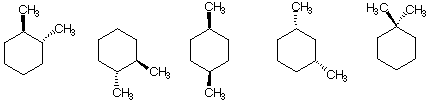
a) The compound(s) with a net dipole moment
![]()
b) The bond length between sp3 hybridized carbons
1.54 D 1.54 x 108 cm 1.09 Å 1.54 Å 1.33 Å
c) The alkane with the lowest boiling point
n-pentane 2-methylbutane n-hexane
cyclohexane 2,2-dimethylpropane
d) Compound(s) containing sp hybridized atoms
CO2 CH2=C=CH2 CH4 BCl3 BeH2
e) Compounds that are deprotonated by NaNH2 (pKa = 33)
CH3CO2H hexane C2H5OH ethylene water
f) The dimethylhexane(s) whose two chair conformations are equal in energy
4) ( 10 pts) Draw a generic structure for each of the following functional groups.
ketone
carboxylic acid
aldehyde
ester
amide
5. (16 pts) On the structure of acrylonitrile. A more elaborate version of this problem appeared on Exam1/97 (cf., PS1/98).
a) (4 pts) Acrylonitrile (1) is used to make polymers. Redraw 1 using lines for bonds to reflect the true geometry of acrylonitrile (be faithful to bond angles; ignore the numbers). In a single word, how would you best describe the shape of acrylonitrile (think of the shapes of ethane, ethylene, and acetylene)?
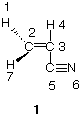
b) (9 pts) Redraw acrylonitrile using lines for s-bonds, correct orbitals for p-bonds and dots for unshared electron pairs.
c) (3 pts) What is the approximate value (degrees) of the bond angle C3-C5-N6 in b)?
6) (12 pts) Bonus question. Match the following luminaries in the history of organic chemistry with their contributions: Baeyer, Kekule/Couper, Lavoisier, Liebig, Dumas/Laurent, van't Hoff/LeBel. Enter the name on the line below..
Theory of combustion __________________
Laboratory research, improved combustion analysis __________________
Tetrahedral carbon _______________________
Tetravalent carbon _________________________
Substitution theory _________________________
Baeyer strain theory ________________________