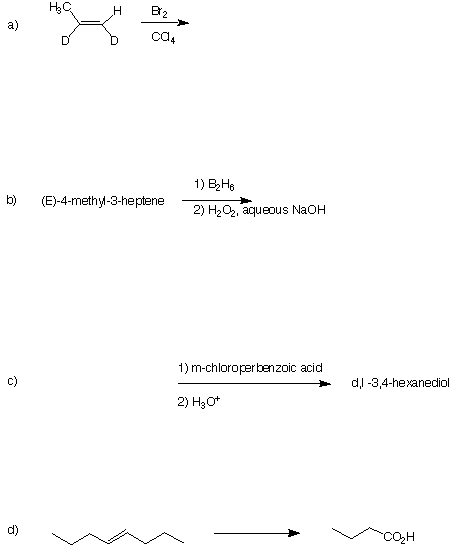
MAKEUP EXAM
CHEMISTRY 220
9: 00 A. M., Wednesday, December 10, 1997
NAME (print): ___________________________________________________
TA:_____________________ Day:_________________ Time:__________
Important points are in bold.
Take a few moments to look over the exam. Do the questions first with which you feel most comfortable. Answer each question on the exam paper.
Do all preliminary drawing or computations on the work sheets at the end of the exam.
There is a Periodic Table of the Elements at the end of the exam.
The exam is one hour.
STOP writing when you are asked to do so.
Reember: Neatness is to your advantage.
1. (20 pts) ______
2. (20 pts) _____
3. (20 pts) ______
4. (20 pts) ______
5. (20 pts) ______
_______________________________
Total (100 pts)
1. (4 x 5 pts = 20 pts) For the radical chain reaction shown below, the first propagation step has a heat of reaction of DHo = -6.6 kcal/mol and the second step has DHo = -26.4 kcal/mol.
2-methylpropane + Cl2 ----------> tert-butyl chloride + HCl
a) What is the first propagation step?
b) What is the second propagation step?
c) What is the heat of the overall reaction?
d) What is the heat of formation of tert-butyl radical given the following data? DHfo (tert-butyl chloride) = -43.3 kcal/mol; DHfo (Cl atom) = +28.9 kcal/mol.
2. (4 x 5 pts = 20 pts) For each of the following reactions, provide the major product (a and b), the starting material (c) or reagents (d). Pay particular attention to stereochemistry. Comment briefly.
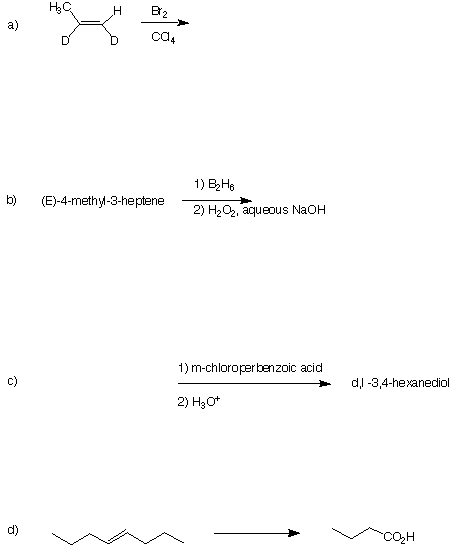
3. (20 pts) An unknown compound A reacts with ozone followed by zinc reduction to give a single compound B. Compound B reveals the following spectral data: 13C NMR (decoupled) d 29 (strong singlet), 204 (weak singlet); 13C NMR (coupled) d 29 (quartet), 204 (weak singlet); 1H NMR d 2.2 (singlet). Explain how the spectral data leads to the structures of A and B.
4. (20 pts) Design a synthesis of 3-hexanone (1) from ethanol and acetylene. All reagents are available to you.
5. (4 x 5 pts = 20 pts) Circle the best answer.
a) The strongest base.
NH3 C2H5ONa CH3CN HCCNa H2O
b) The compound with DGo=0 for the energy between its cyclohexane chair conformations.
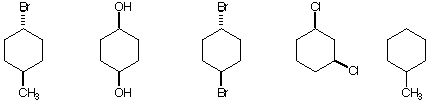
c) The compound not at the same oxidation level as the other four.
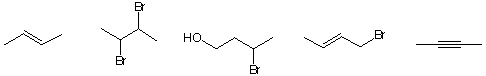
d) The IUPAC name for ![]()
(2S),(Z)-2,4-dibromo-3-hexene (5S),(Z)-3,5-dibromo-3-hexene
(2R),(Z)-2,4-dibromo-3-hexene (2R),(E)-2,4-dibromo-3-hexene
(5S),(E)-3,5-dibromo-3-hexene