EXAM 2
CHEMISTRY 220
Friday, October 17, 1997
NAME (print): ___________________________________________________
TA:_____________________ Day:_________________ Time:__________
Important points are in bold.
Take a few moments to look over the exam. Do the questions first with which you feel most comfortable. Answer each question on the exam paper.
Do all preliminary drawing or computations on the work sheets at the end of the exam. There is a Periodic Table of the Elements at the end of the exam.
The exam is 55 minutes.
STOP writing when you are asked to do so.
REMEMBER: Neatness is to your advantage. 1. (20 pts) ______
2. (10 pts) ______
3. (20 pts) ______
4. (20 pts) ______
5. (12 pts) ______
6. (18 pts) ______
________________________________
Total (100 pts)
1. (20 pts) In class we discussed the free radical chlorination of methane to form methyl
chloride and hydrogen chloride. One can conceive of the following radical chain
reaction to account for the products. However, this reaction sequence does not
take place.
1) Cl. + CH4 ------> CH3Cl + H.
2) H. + Cl2 ------> HCl + Cl.
a) (6 pts) Given the following bond dissociation energies, calculate the heat of reaction of
each of the propagation steps above and the heat of the overall reaction.
DHo (kcal/mole): CH4 = 105; CH3Cl = 85; HCl = 103; Cl2 = 58. Show work.
step 1 ----->
step 2 ----->
overall reaction ------->
b) (10 pts) Write the correct propagation steps for the free radical chain reaction of
methane with chlorine. Calculate the heat of each step and the heat of the overall
reaction. Show work.
step 1 ----->
step 2 ----->
overall reaction ------->
c) (4 pts) Briefly explain why the radical chain of part b) is operative and the radical chain
of part a) is non-operative.
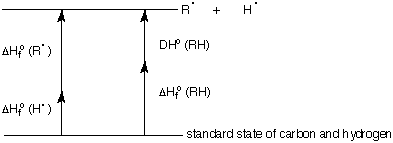
2) (10 pts) The diagram below illustrates the relationship between heats of formation and
bond dissociation energy for a hypothetical hydrocarbon.

a) (5 pts) Write an equation that solves for DHfo (R.).
b) (5 pts) Compute the heat of formation of ethyl radical from the following data
(kcal/mole) using your equation of part a):
DHfo (ethane) = -20.2; DHo (ethane) = 101; DHo (H2) = 104. Show work. [Note:
There is no specific value given for DHfo (H.).]
3. (4 x 5 = 20 pts) Circle the correct answer(s) in the following:
a) The compound(s) that can exist as enantiomers:
3-pentanone 2-pentanone cis-1,3-dichlorocyclopentane 2-octanol 3-octanol
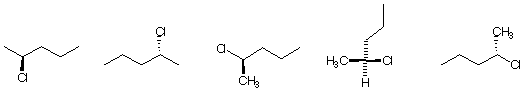
b) The structure(s) that represent (R)-2-chloropentane:
c) The compound(s) that have a net dipole moment:
1,2-dichloroethane H2O hexachloroethane chloroform methyl bromide
d) The atom(s) or molecule(s) whose heat of formation ( DHfo) = 0 at 25 oC:
O2 N2 C(diamond) Cl2(gas) Br2(gas) I2(gas)
4. (20 pts) Both enantiomers of compound A, C7H12NCl, were prepared
in unequal
amounts in a laboratory. Prior to recording their rotations, the samples were
inadvertently mixed together. By an independent method, the composition of the mixture was determined to be 4:1. The observed optical rotation of the mixture was -66o.
a) (6 pts) How many degrees of unsaturation (rings and multiple bonds) are present
in A?
b) (8 pts) What is the specific rotation of the major enantiomer? Show work.
c) (3 pts) What is the specific rotation of the minor enantiomer?
d) (3 pts) What is the configuration (R, S) of the major
enantiomer?
5. (12 pts) The fat soluble anti-oxidant, vitamin E (a-tocopherol) is shown below.
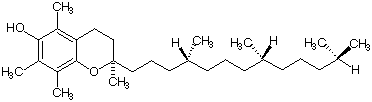
a) (6 pts) How many additional stereoisomers of vitamin E are there?
b) (6 pts) Indicate the R,S configuration at each of the
stereogenic centers in vitamin E. [Show above].
6. (18 pts) Two compounds A and B have the molecular formula C5H11Cl and they are
both of the (R)-configuration. Each one undergoes the sequence of reactions shown
below. Both D and F have the formula C5H11D. D and F are racemic. Draw the
structures for A-F where indicated and specify the configuration for A and B. [Note: A can be B and B can be A. Hint: What are the three different ways a C5 chain can be arranged and where can you place the chlorine atom in each one?]
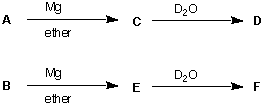
A
B
C
D
E
F