EXAM 3
CHEMISTRY 220
Friday, November 8, 1996
NAME (print): ___________________________________________________
TA:_____________________ Day:_________________ Time:__________
Take a few moments to look over the exam. Important points and unknowns are in bold type.
Answer each question on the exam paper. Do all preliminary drawing or computation in a blue
book. Blue books will not be collected. The exam is 55 minutes. The points should be an
indication of the time required for solution. STOP writing when you are asked to do so.
REMEMBER: Neatness is to your advantage.
1. (25 pts) ______
2. (24 pts) ______
3. (18 pts) ______
4. (16 pts) ______
5. (17 pts) ______
--------------------------------------------------------------------
TOTAL (100 pts) ______
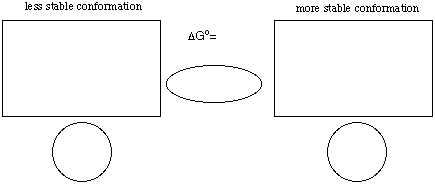
1. (25 pts) For Your Information. The chair conformation of bromocyclohexane bearing
an equatorial bromine is 1.0 kcal/mol more stable than the chair conformation having an
axial bromine.
a) (3 pts) Name the two stereoisomers of 1,4-dibromocyclohexane.
b) (3 pts) What is the relationship of these stereoisomers to one another?
c) (3 pts) Can they be resolved into enantiomers? Explain briefly.
d) (10 pts) Draw in the rectangles the 3D structures of the chair comformations of the
stereoisomer of 1,4-dibromocyclohexane whose KeqŁ1. Be sure that you place the
correct structure in the correct box. Place equilibrium arrows indicating the direction
of the equilibrium in the oval.

e) (3 pts) Calculate the energy of each conformation and place the answer in the circle
below the appropriate structure.
f) (3 pts) Calculate DGo and place the value next to DGo= in part d).
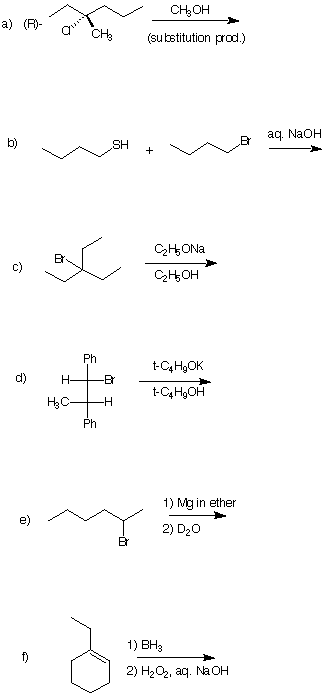
2. (6 x 4 pts= 24 pts) Predict the major product of each of the following reactions. Be
explicit about stereochemistry, racemization, etc. Comment briefly if necessary.
3. (18 pts) There are four stereoisomers (two pairs of enantiomers) of 5-bromo-2-hexanol
(1). When 1 reacts with potassium tert-butoxide, 2,5-dimethyltetrahydrofuran (2) is
formed as a substitution product by an SN2 mechanism.
a) (6 pts) If compound 2 is the (R,R)-stereoisomer, draw the correct stereoisomer of 1
that produces 2 and label the centers of stereochemistry in 1 with R and S.
b) (6 pts) Provide a mechanism for 1 ---> 2 that clearly illustrates (3D structure) the
stereochemistry of the reaction in part a). Include the role of potassium tert-butoxide.
c) ( 6pts) Name the other stereoisomer of 1, beside (2S, 5S)-5-bromo-2-
hexanol (1)that will provide meso-2,5-dimethyltetrahydrofuran 2.
4. (16 pts) (E)-1-Trimethylsilyl-1-butene (1) reacts with Br2 to form a single,
diastereomerically-pure dibromide (2). When 2 reacts with sodium methoxide, (Z)-1-
bromo-1-butene (3) is formed along with methoxytrimethylsilane. Demonstrate your
knowledge of the mechanism of the Br2 addition to double bonds (1 ---> 2) and of
the E2 elimination reaction (2 ---> 3). Use 3D structures that clearly tell the
mechanistic story (use arrows). [Hint: The trimethylsilyl group may be treated as a
"big b-hydrogen atom".]
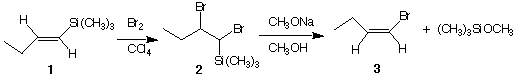
5. (17 pts) Compound A (C7H12) reacts with ozone (followed by dimethyl sulfide reduction)
to give only 3-methylhexandial (3-methylhexane dialdehyde) B. Compound A reacts with
OsO4 to give two diastereomeric products, C and D (C7H14O2). Draw the structures of
A-D. Provide stereochemistry where appropriate.