MAKE-UP
EXAM
CHEMISTRY 220a
Friday, December 13, 2002
NAME (print): ______________________________________________________________
TA:_____________________ Section Day:_________________ Section Time:__________
The exam is 55 minutes; it covers Chapters 1-11.
Take a few moments to look over the exam. Answer each question on the exam paper.
Important clues and instructions are in bold.
Do all preliminary drawing or computations on the work sheets at the end of the exam. The work sheets will not be graded. There is a Periodic Table on the last page of the exam.
STOP writing and hand in your exam when you are asked to do so.
Remember: Neatness is to your advantage.
1. (20 pts) ______
2. (20 pts) ______
3. (20 pts) ______
4. (20 pts) ______
5. (20 pts) ______
_______________________________
Total (100 pts)
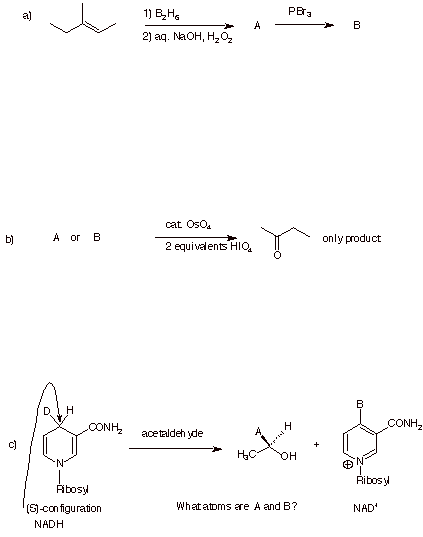
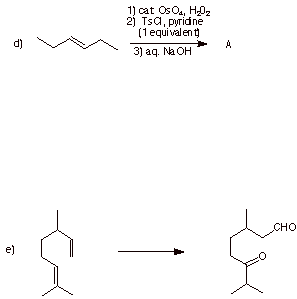
1. (20 pts.) Do 4 of 5. Provide the missing information in each of the following reactions. Pay attention to stereochemistry.
2. (20 pts.) Two enantiomers have equal and opposite specific rotations of 120o. If a mixture of the two enantiomers has a specific rotation of +80o, How much (percent) of the dextro- and laevorotatory enantiomers are present? What is the optical purity of the mixture? Show work.
3. (20 pts.) An overall chemical reaction (A à C) having an intermediate B is exothermic by 20 kcal/mol and the first step is endothermic by 3 kcal/mol. Illustrate this reaction with an energy diagram taking into account the Hammond Postulate. Indicate the rate limiting step in the reaction? What is DHo for the second step?

4. (20 pts.) Design a synthesis of 3-ethyl-2-pentene (C7H14) from ethylene and formaldehyde as your source of carbon atoms. All reagents are available to you. [Note: Ethylene has an even number of carbons; C7H14 has an odd number.]
5. (20 pts.) Ozonolysis and dimethyl sulfide reduction of alkene A (C7H14) provides B and C. Mercuric sulfate catalyzed hydration of 1-pentyne affords only D while hydration of 2-pentyne gives C and D. What are the structures A-D? Explain.
Work Sheets
Work Sheets
Work Sheets
Periodic Table