EXAM 3
CHEMISTRY 220a
Friday, November 8, 2002
NAME (print): ___________________________________________________
TA:_____________________ Day:_________________ Time:__________
Take a few moments to look over the exam. Answer each question on the exam paper.
Important clues, points, and structures are in bold.
Do all preliminary drawing or computations on the work sheets at the end of the exam. The work sheets will not be graded
There is a Periodic Table on the last page of the exam.
The exam is 55 minutes.
STOP writing and hand in your exam when you are asked to do so.
Remember: Neatness is to your advantage.
1. Structure (20 pts) ______
2. Synthesis (20 pts) ______
3. Reactions (30 pts) ______
4. Potpourri (30 pts) ______
_______________________________
Total (100 pts)
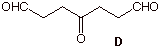
1. Structure: (20 pts.) Compound A (C9H14) reacts with two moles of H2 in the presence of Pd or Pt to form B. Compound B is found to be not identical with 1-cyclobutylpentane. Ozonolysis of A and subsequent reduction with dimethyl sulfide affords keto dialdehyde D and compound E. What are the structures A-E? Explain and illustrate.
2. Synthesis: (20 pts.) A chemist requires a sample of racemic 4,5-octanediol (C8H18O2). She designs and executes a synthesis of the diol using acetylene (C2H2) and propene (C3H6) as her sources of carbon. All other reagents were available to her. Show how she may have accomplished her goal.
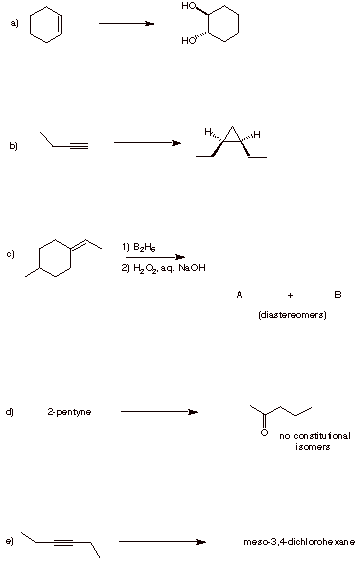
3. Reactions: (30 pts.) In each of the following chemical transformations, provide the reaction conditions or the products. Several steps may be required. Pay attention to stereochemistry.
4. Potpourri: (30 pts.) Circle the best answer(s) in each of the following.
a) The reactions that are conducted in the presence of a catalytic reagent.
Hg++ hydration of acetylenes dihydroxylation with OsO4/H2O2
anti-Markovnikov addition of Lindlar reduction
HBr to alkenes
hydroboration
b) Racemates are expected in the reaction of (Z)-5-decene with
Br2 Br2/H2O peracid OsO4/H2O2 CH2I2/Zn(Cu)
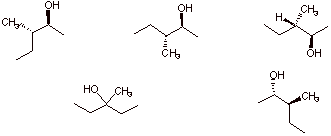
c) The structure(s) that represent the correct stereochemistry from the hydroboration of (E)-3-methyl-2-pentene.
d) The compound(s) in violation of Bredt’s Rule

e) The compound(s) with the smallest heat of hydrogenation

Work
Sheets
Work Sheets
Periodic
Table