CHEMISTRY 220a
Friday, September 28, 2001
NAME (print): ___________________________________________________
TA:_____________________ Day:_________________ Time:__________
Take a few moments to look over the exam. Answer each question on the exam paper.
Important clues and structures are in bold.
Do all preliminary drawing or computations on the work sheets at the end of the exam. The
work sheets will not be graded. There is a Periodic Table on the last page of the exam.
The exam is 55 minutes.
STOP writing and hand in your exam when you are asked to do so.
REMEMBER: Neatness is to your advantage.
1. (20 pts) ______
2. (20 pts) ______
3. (20 pts) ______
4. (20 pts) ______
5. (20 pts) ______
_________________________
Total (100 pts)
1. (20 pts) Compound A, C7H14O, is a 1,3-disubstituted cyclohexane. The difference in
energy between its two chair conformations is 0.7 kcal/mol. [DGo (kcal/mol) values for
axial and equatorial substituents in mono-substituted cyclohexanes: -CN, 0.2; -Cl, 0.5;
-OH, 1.0; -COOH, 1.4; -CH3, 1.7; -C2H5, 1.8.]
a) (6pts) What are the two groups attached to the cyclohexane ring? Briefly explain yourreasoning.
b) (6pts) Is this 1,3-disubstituted cyclohexane a cis or trans isomer? Explain briefly.
c) (8pts) Using the template below, add the groups to the cyclohexane rings paying
attention to the position of the equilibrium. Briefly explain your reasoning.

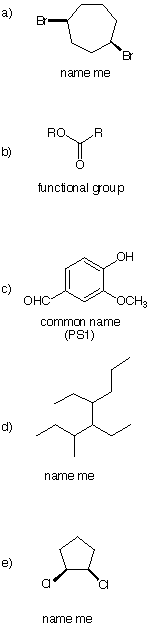
2. (20 pts) Vinyl acetylene 1, is an important intermediate in the preparation of synthetic
rubber. Draw a molecular orbital representation of 1. Include and indicate the
hybridization of carbon atoms, the location of p-bonds, and an overall description (in a
single word) of the shape of the molecule.
3. (20 pts; equal weight) Circle the best answer(s) in each of the following questions.
a) The dihedral angle in gauche butane
|
|
|
|
|
|
b) Compounds with a net dipole moment
![]()
c) The least acidic of the following compounds
|
|
|
|
|
|
d) The 19th century chemist who concerned himself with stability of cycloalkanes.
|
Baeyer |
|
|
|
|
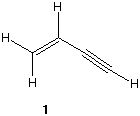
e) The dibromocyclohexanes that have two equatorial substituents in their most stable chair
conformation.
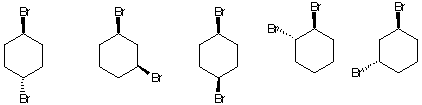
4. (20 pts) For the eclipsed and staggered conformations of 2-methylpentane viewed along the
C2-C3 sigma bond, draw a Newman projection of the most stable eclipsed and staggered
conformations. Place the appropriate energies in the Newman projections below. [Use the
circles as templates for the Newman projections.] Calculate the energy (kcal/mol) of both
conformations. Place your answer in the appropriate box. Show work. [H/H, eclipsed, 1.0
kcal/mol; CH3/H eclipsed, 1.3 kcal/mol; C2H5/H, eclipsed, 1.4 kcal/mol; CH3/CH3, eclipsed,
3.0 kcal/mol; CH3/CH3, gauche, 0.9 kcal/mol;
CH3/C2H5, gauche, 1.0
kcal/mol.]
5. (20 pts) Complete the following: