FINAL EXAM
Organic Chemistry
Chemistry 220a; 2 P.M., Wednesday, December 19, 2001
NAME (print): ______________________________________________________________
TA:_____________________ Section Day:_____________ Section Time:______________
Take a few moments to look over the exam. Do problems first with which you are most comfortable. Important points and unknowns are in bold type. Do all preliminary work on the worksheets. The worksheets will not be graded. The following tables are at the end of the exam: Periodic Table (page 21), BDEs (page 22), Common Isotopes (pg. 23). The exam is 2 to 2-1/2 hours with an additional 1/2 hour for review. STOP writing when you are asked to do so. Put your name on the cover sheet and subsequent pages where indicated.
For questions 2 and 4, you are to complete only one of the two choices that are offered for each.
For question 3, do 4 of 6. See directions on the exam.
For question 5, do 1 of 4.
For question 6, do 4 of 6.
For question 8, do 5 of 6.
Remember: Neatness is to your advantage. Have a GREAT winter
break! See you next term for Chem
221b.
1. Naproxen (36 pts) ________ 5. Mechanism (20 pts.) _________
Revisited
2. Structure (30 pts) _________ 6. Kinetics/ (32 pts.) _________
Thermodynamics
3. Potpourri (40 pts) _________ 7. Radicals (32 pts.) _________
4. Synthesis (30 pts) _________ 8. Reactions (30 pts.) _________
______________________________________________________________________
Total (250 pts) ______________
1. Naproxen Revisited: (6 x 6 pts. = 36 pts.) In exam 2, we considered the resolution of racemic Naproxen to isolate (S)-(+)-Naproxen (A). The current method for preparing (S)-(+)-Naproxen is by catalytic hydrogenation of carboxylic acid B.
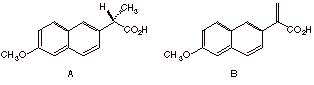
a) Is B chiral or achiral? Why?
b) Will hydrogenation of B in the presence of a noble metal such as Pt form (S)-(+)-Naproxen efficiently (>75%)? Explain briefly.
c) The optically active rhodium catalyst C, Rh(DIOP)Cl2, which was used in your text (pg. 219) to illustrate asymmetric hydrogenation [2001 Nobel Prize in Chemistry], might be an ideal catalyst to produce only a single enantiomer of Naproxen. Briefly illustrate and/or explain the principle of asymmetric hydrogenation in general terms. [Hint: Recall how you were to illustrate the resolution of Naproxen in exam 2.]
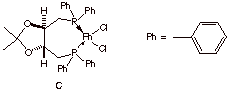
d) What are the configurations (R/S) of the
stereocenters present in the ligand D used in the formation of catalyst C?
Label them below.
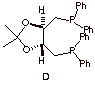
e)
The four carbon chain of the ligand D above is derived from one of the stereoisomers of
tartaric acid. Which one is it?
Draw it in a Fischer projection.
[Hint: see below]

f) The tartaric acid is
converted in three steps to compound E (X = a good leaving group).
What type of mechanism is occurring in the conversion E ---> D?
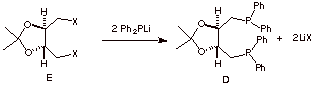
g)
Why is the asymmetric hydrogenation approach inherently more efficient
than the resolution strategy? Be brief.
Do only 2a or 2b, not both of them!
2a. Structure: (30 pts.) Alkene A, C10H20, reacts with OsO4/H2O2, to give (±)-diol B. This substance reacts with periodic acid to produce a single compound C, C5H10O. Compound C can be synthesized from 1-pentyne in a single chemical reaction. Compound C is inert toward aqueous chromic acid but it reacts with NaBH4/H2O or LiAlH4/ether to form D, C5H12O.
Place an "X" here if you want 2a graded. ______
i) (25 pts.) What are the structures of A - D? Briefly justify your answers.
ii) (5 pts.) The mass spectrum of D is shown below.
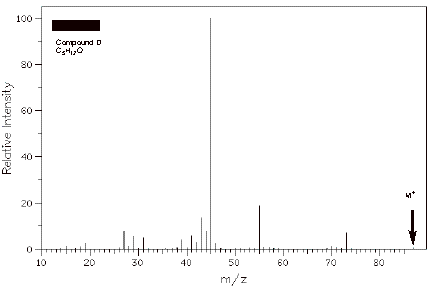
What is the value of m/z for the base peak?
What is the value of m/z for the molecular ion?
Do only 2a or 2b, not both of them!
2b. Structure: (30 pts.) Ester A, C6H12O2, reacts with two equivalents of RMgX (compound B) to provide alcohol C (C4H10O) and alcohol D (C8H18O). Alcohol C is converted into E (C4H8O) by either PCC/CH2Cl2 or aqueous chromic acid. Treatment of alcohol D with H2SO4 leads to compounds (E)-F, (Z)-G, and H, all three of which have the formula C8H16. Compound H is the least abundant of the three. The reaction of compound H with O3 and then (CH3)2S permits the ready isolation of 4-heptanone. What are the structures of A-H? Briefly justify your answer.
Place an "X" here if you want 2b graded. ______
Do
3a and 3b and two of the remaining four questions (3c-f).
3. Potpourri: (4 x 10 pts. = 40 pts.) If you manage to do 5 or 6 problems, cross out the one(s) of 3c-f that you do not want graded.
a) Three compounds, A, B, and C, have the formula C5H10 and all three of them afford 2-methylbutane upon catalytic hydrogenation. The three compounds have the following heats of formation (kcal/mol): A, -6.8; B, -8.4; C, -9.9. What are the structures of A, B, and C. Illustrate and explain briefly.
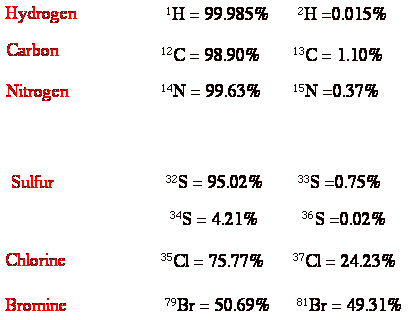
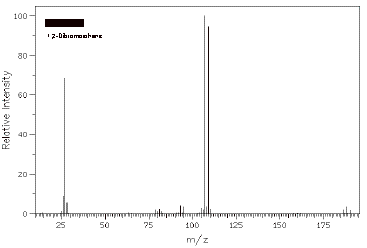
b) 1,2-Dibromoethane has three peaks for its molecular ion(s) of relative intensity 1:2:1. What are the masses of these 3 peaks and why are their relative intensities what they are?
(Common Isotopes, pg. 23)
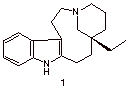
c) The indole alkaloid quebrachamine (1) has a specific rotation [a]D = -110o. An attempted resolution of (±)-1 led to a sample that had [a]D = -33o. What percentage of the dextrorotatory enantiomer was present? Show work.
d) The peracid epoxidation of (E)-2-butene (zero inversions) is the operational equivalent of what two inversion process? Illustrate and explain briefly.
e) What is one property that the compounds acetylene, hydrogen cyanide, and BeCl2 share in common? Explain briefly.
f) Arrange (left to right) the following carbocations in increasing order of stability: (CH3)3C+ (t-butyl cation), (CH3)2CH+ (2-propyl cation), and CH3OC(CH3)2 (2-methoxy-2-propyl cation). Explain briefly.
Do only 4a or 4b, not both
of them!
4a. Synthesis: (30 pts.) A chemist requires a sample of 2,5-dimethyl-2-hexanol (1, C8H18O). She recognizes an element of symmetry (C4 + C4) in the hydrocarbon structure of the alcohol. She designs and executes a synthesis of 1 from acetone and methanol. She has all reagents and solvents available to her as do you. How would you complete the synthesis of this alcohol under the restrictions indicated?
Place an "X" here if you want 4a graded. ______
Do only 4a or 4b, not both
of them!
4b. Synthesis: (30 pts.) A chemist isolates an optically active compound from nature which he believes to be (Z)-3-methyl-4-heptene-3-ol (1, C8H16O). He synthesizes 1 from 2-butyne as his only source of carbon. All reagents and solvents are available to you as they were to him. Reconstruct his synthesis.
Place an "X" here if you want 4b graded. ______
Is the
synthetic alcohol optically active? Justify.
5. Mechanism: (20
pts.) Provide the mechanism of one,
and only one, of the following reactions. Use the curved arrow formalism.
The Na/NH3 reduction of 2-butyne
The ozonolysis/dimethyl sulfide reduction of (E)-2-butene
The hydroboration/oxidation of (E)-3-methyl-2-pentene
The reaction of HBr in the presence of a peroxide with 2-methyl-2-butene
6. Kinetics/Thermodynamics: (4 x 8 pts. = 32 pts.) Do 4, and only 4, of the following 6 problems. If you manage to do 5 or 6 problems, cross out the one(s) that you do not want graded. If structures are shown, circle the correct answer and provide a few words of justification. If no structure is shown, give a brief explanation.
a) The compound with the lower rate for E2 elimination. Justify.
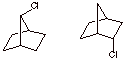
b) The compound with the greater rate of
E2 elimination. Justify.

c) Of (Z)-2-butene and (E)-butene, the one with the greater heat of hydrogenation. Illustrate and justify.

d) The compound that gives the higher ratio (SN2/E2) of SN2 to E2 product when reacted with KOH. Justify.
e) The heat of hydrogenation of ethylene to ethane is -32.9 kcal/mol and the heat of hydrogenation of acetylene to ethane is -74.5 kcal/mol. What is DHfo for ethylene given that DHfo for acetylene is +54.3 kcal/mol.
f) The C-H bond in acetylene is stronger than tthe C-H bond of ethane yet the C-H bond in acetylene is ~25 orders of magnitude more acidic than that in ethane. Explain briefly.
7. Radicals: (32 pts.) Benzylic C-H bonds, like allylic C-H bonds, have low bond dissociation energies relative to alkane C-H bonds (see page 22, BDEs ). The free radical chlorination of toluene (PhCH3, 1) gives benzyl chloride (PhCH2Cl, 2). The second propagation step has DHo = -10 kcal/mol.
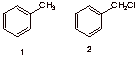
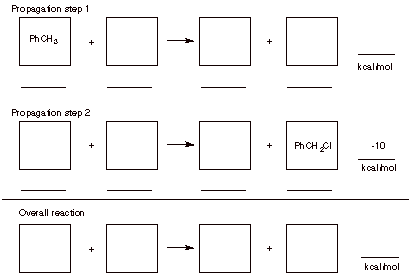
a) (12 pts.) Write the propagation steps for this reaction. Place the structures in the boxes and the BDEs on the appropriate lines. Determine the heat of reaction for the 1st propagation step. Put your answer on the appropriate line.
b) (8 pts.) Determine the overall heat of the reaction. Place your answer in the designated place.
c) (12 pts.) Given the following values for DHfo
(kcal/mol): PhCH3 =
+12; HCl = -22, determine the heat of formation (DHfo) of PhCH2Cl.
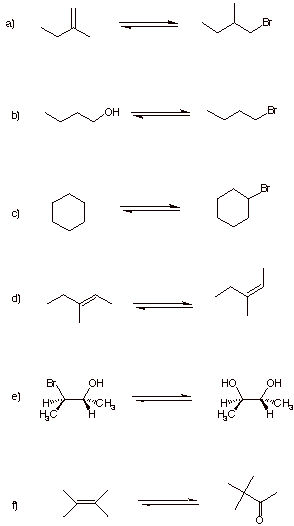
8. Reactions: ( 5 x
6 pts. = 30 pts.) Do 5, and only
5, of the following 6 problems. If you manage to do 6 problems, cross
out the one that you do not want graded.
Provide the reagent(s) for the forward reaction (above the arrows) and
for the reverse reaction (below arrows).
Each reaction requires one or two steps. [Note: The arrows do not indicate equilibria.]