Organic Chemistry
Chemistry 220a
2 P.M., Friday, December 15, 2000
NAME (print): ______________________________________________________________
TA:_____________________ Section Day:_____________ Section Time:______________
Take a few moments to look over the exam. Do problems first with which you are most
comfortable. Important points and unknowns are in bold type. Do all preliminary work on
the worksheets. The worksheets will not be graded. There is a Periodic Table (page 19) and a
table of BDEs (page 20) of the exam. The exam is 2 to 2-1/2 hours with 1/2 hour for review.
STOP writing when you are asked to do so. Put your name on the cover sheet and subsequent
pages where indicated.
REMEMBER: Neatness is to your advantage. Have a GREAT winter break! Best of
Luck in Chem. 221b!
1. (30 pts) Structure/NMR _______________
2. (40 pts) Potpourri _______________
3. (40 pts) Radicals ________________
4. (30 pts) Reactions ________________
5. (20 pts) Kinetics ________________
6. (40 pts) Structure ________________
7. (30 pts) Synthesis/NMR ________________
8. (20 pts) Mechanisms ________________
_________________________________________________________
Total (250 pts)
1) Structure/NMR: (30 pts) The terpene, (R)-citronellal ([a]D = +12.0o) A, which is a
constituent of citronella oil, contains two degrees (elements) of unsaturation and it has the
broad band decoupled 13C NMR spectrum shown below. [Note the following: i) Examine
carefully the "two" signals in the region between 25-30 ppm. ii) There is more than enough
information in the problem to determine the molecular formula of A.]
Ozonolysis of citronellal and subsequent reaction with dimethyl sulfide provides compounds B
and C. The 1H NMR spectrum of B displays a singlet only (d 2.2). The 13C NMR broad
band decoupled spectrum of B displays two singlets (206 and 30 ppm). Compound C,
(C7H12O2) reacts with NaBH4 to produce (R)-3-methylhexan-1,6-diol D. Upon reaction of
citronellal with hydrogen in the presence of a palladium catalyst, compound E is formed. The
reaction of E with LiAlH4 affords compound F, (3R)-3,7-dimethyloctanol.
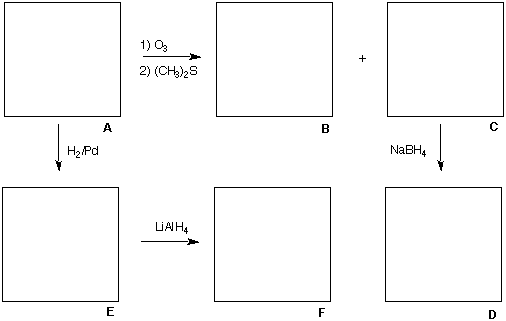
What are the structures of A-F? Place your answers in the appropriate boxes and give a brief
rationale below. Pay attention to absolute stereochemistry.
2) Potpourri: (8 x 5 pts. = 40 pts.) Provide the best answer(s) in each of the following
questions:
a) The value of a particular coupling constant (J) is 8 Hz at 60 MHz. What will be the value
of J at 300 MHz? (Enter your answer.)
b) The hybridization of carbons in the allene, (S)-2,3-pentadiene. (Mark your ballot. No
hanging chads will be accepted.)

c) The value (kcal/mole) of D(DGo) for the equilibrium between the chair conformations of
trans-1,3-dimethylcyclohexane. (Circle your answer.)
|
0.9 |
1.8 |
2.7 |
3.6 |
none of these |
d) The acid that has the median pKa. (Circle your answer.)
|
acetic acid |
ethanol |
t-butyl alcohol |
ammonia |
methane |
e) The dichloro bicyclo[2.1.1]hexane(s) that displays four singlets in its broad band
decoupled 13C NMR spectra. (Circle your answer.)

f) A sample of citronellal (problem 1) that has [a]D = -9.0, contains what percentage of the
(S)-enantiomer? (Circle your answer.)
|
12.5 |
75 |
25 |
50 |
87.5 |
g) For a series of sp3, sp2, and sp C-H bonds, the bond dissociation energies (BDEs) and
pKa are related how? (Circle your answer.)
|
linearly |
exponentially |
logarithmically |
inversely |
h) The bond (bold) with the largest bond dissociation energy (BDE). (Circle your answer.)

3) Radicals: (40 pts)
a) (10 pts) For the free radical iodination of ethane, provide the propagation steps and the
overall reaction for this process in the gas phase. Place the structures in the appropriate boxes
below. BDEs
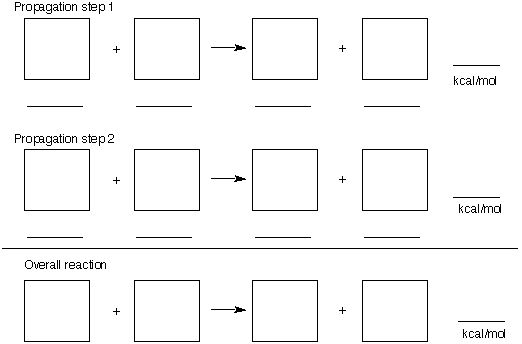
b) (10 pts) Compute the thermicity of the three reactions above placing the appropriate
answers and BDEs (page 20) on the lines under the boxes. Show your work here. Why is
this procedure an ineffective method for preparing alkyl halides?
c) (10 pts) Iodine is a solid at 25 oC and 1 atm. Using the following data and your answer in (b), determine the heat of vaporization (sublimation) of I2 at 25 oC and 1 atm: DHfo
(kcal/mol); C2H6 = -20.0, C2H5I = -1.9, HI = +6.3. Show work.
d) (10 pts) Provide an energy vs. reaction coordinate diagram using the diagram below.
Label the energy differences (values obtained in part b), be true to the Hammond Postulate,
and the height of the transition states.

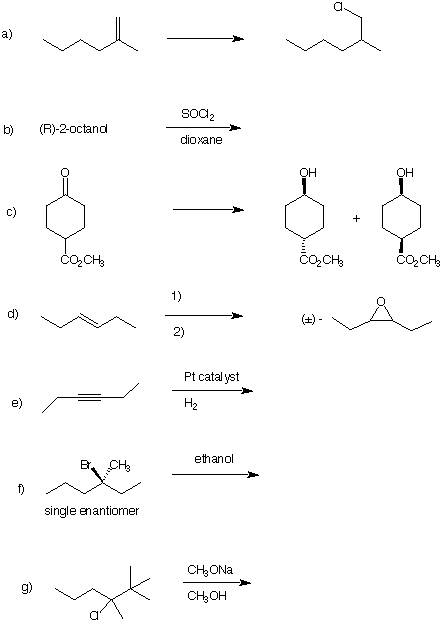
4) Reactions : (6 x 5 pts = 30 pts) Do 6 of the following 7 problems. Place an "X"
through the problem that is not to be graded. Some questions require products; some
reagents. Pay attention to stereochemistry.
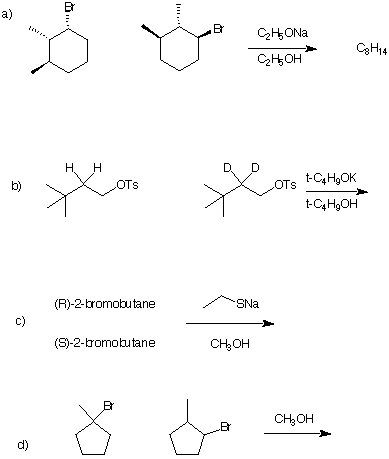
5) Kinetics: (4 x 5 pts = 20 pts) In each of the following problems, circle the compound that
reacts faster and draw the structure of the product of the reaction. Give a brief
explanation. [Note: If two reactants react at the same rate, circle both of them and clearly
specify the product.]
|
6) Structure: (40 pts) (R)-(+)- Piperitone (1) is a terpene with the fragrance of peppermint. The d-form is reported to have [a]D = 49 o while the l-form is reported as [a]D = -16 o. A sample of piperitone from a bottle in the FEZ laboratory had [a]D = -40o. |
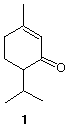 |
a) (10 pts) Assuming that enantiomerically-pure piperitone has [a]D = 50o, calculate the
%ee of the FEZ sample. Show work.
b) (10 pts) Using your knowledge of keto-enol tautomerization, briefly explain how the two
l-forms cited above became partially racemized. Be brief and concise.
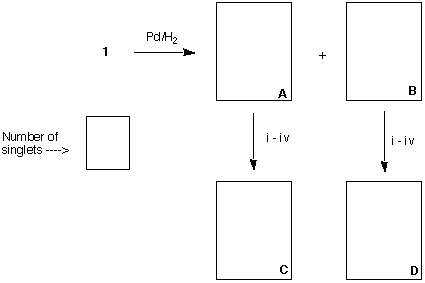
c) (20 pts) Hydrogenation of piperitone in the presence of a Pd catalyst provides ketones A
and B (both C10H18O). Ketone A provides C (C10H20) and ketone B affords D
(C10H20) after the following series of reactions: i) NaBH4 or LiAlH4; ii) PBr3; iii) Mg in
ether; iv) H2O. Compound C has a D(DGo) = 0.3 kcal/mol between its two chair
conformations while compound D has D(DGo) = 3.9 kcal/mol between its two chair
conformations. Both C and D contain the same number of singlets in their respective
broadband decoupled 13C NMR spectra. Provide structures for A-D aand the number of
singlets in the 13C NMR spectra. Explain briefly.
|
7) Synthesis/NMR: (30 pts) A chemist requires a sample of 2,2-dideuterio-3- methyl-1-butanol (1). She designs a synthesis of 1 from isobutylene. She has available to her all C1 compounds and all reagents including NaBD4, LiAlD4 and D2O. |
|
a) (15 pts) Show how she may have -- or you would -- accomplish this synthesis. [Note:
Not all deuterium reagents are necessary for the completion of the synthesis.]
b) ( 15 pts) Use the template below to draw the 1H NMR spectrum of 1 that has the
hydroxyl hydrogen exchanged with D2O. [Note: Since deuterium bound to oxygen does not
couple to vicinal hydrogens, you may make the same assumption about carbon bound
deuterium.] Place the signals in their correct, relative positions (chemical shifts). Include the
coupling patterns and the relative integrals.
![]()
8) Mechanisms: (20 pts) Using the curved arrow formalism, provide a mechanism for one
and only one of the following reactions.
a) 2-butyne ------> (E)-2-butene
b) isobutylene ------> isobutyl alcohol (2-methyl-1-propanol)
c) (Z)-2-butene ------> CH3CHO
d) C2H5OH ------> CH3CHO