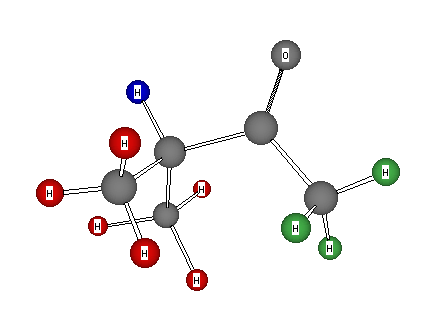
Formula: C5H10O
Answer: 3-Methyl-2-butanone (Isopropyl methyl ketone)
1H Chemical Shift Assignments: δ 1.05 (s, 6H), 2.09 (s, 3H), and 2.55 (1H, septuplet)
This spectrum reveals an isolated isopropyl and methyl group. The sum of these two groups (C4H10) leaves C=O to separate the two groups from coupling to one another. Note the lower field chemical shift of the methine hydrogen vs. the methyl group. The methylene group in 3-pentanone absorbs in between these values (δ 2.37).
The 13C spectrum displays a weak signal at 210.5 ppm due to the C=O, which has no attached hydrogens to assist in relaxation. The signals at 44.6 and 29.5 ppm are the carbons attached to the C=O while the methyl groups of the isopropyl group are identical and are located at 17.1 ppm. Return to Menu.
 |
oxygen-orange
|
|---|