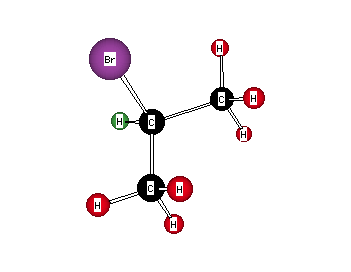
Formula: C3H7Br
Answer: 2-Bromopropane (isopropyl bromide)
1H Chemical Shift Assignments: δ 1.64 (d, 6H), 4.24 (septuplet, 1H)
The two methyl groups are enantiotopic and necessarily equivalent. The 13C spectrum has two singlets: 28.6 and 45.2 ppm. There are only two types of carbon. The six protons at δ 1.64 split the methine proton at δ 4.24 into a septuplet (J = 8.0 Hz). Conversely, the methine proton splits the methyl groups into a doublet (J = 8.0 Hz). This is a typical isopropyl pattern. Compare the 13C NMR spectra of isopropyl bromide and isopropyl iodide here. Return to Menu.
 |
bromine-violet |
|---|