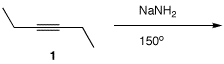
Alkyne 6:
Problem
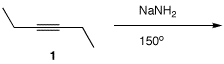
Solution

Internal (disubstituted) alkynes are reversibly isomerized with catalytic NaNH2 at elevated temperatures. Thus, 3-hexyne is isomerized to 2-hexyne and visa versa through the intermediacy of the allene, 2,3-hexadiene. 2-Hexyne can also be isomerized to 1-hexyne (pKa = 25), which is deprotonated by NaNH2 (NH3; pKa = 35). Because of this irreversible step, stoichiometric quantities of NaNH2 must be used. The same reaction may be accomplished with fused KOH at 200oC. Under these conditions all three hexynes are in equilibrium because KOH is not basic enough to effect deprotonation. For a mechanism of the isomerization, click here.