1. When ethanethiol and n-butyl bromide are added to aqueous KOH, n-butyl ethyl sulfide is
formed. Explain.
2. When (R)-bromopropionic acid is exposed to aqueous KOH, (R)-lactic acid is formed.
Explain.
3. Two of the four optically active stereoisomers of 4-chloro-3-hexanol form an optically
inactive compound A, C6H12O, when treated with aqueous KOH. Which ones are they?
What is the structure of A? [Draw their structures, use R,S designations, and provide a
mechanism and explanation for the process.
4. The stoichiometric reaction of ammonia with n-butyl iodide can be used to prepare n-
butylamine. Show a mechanism for this reaction and explain how the free amine, not its
salt, can be isolated.
5. Unfortunately, the reaction described in #4 has a complication. Di-n-butylamine is also
formed. Explain its formation.
6. What change(s) in the reaction conditions of #4 can be made to repress the formation of di-
n-butylamine?
7. (2R,4R)-4-bromo-4-methyl-2-hexanol gives two optically active diols when exposed to water.
However, (R)-3-bromo-3-methylhexane gives racemic alcohol under the same conditions.
Explain.
8. Show how (2R,5R)-2,5-dimethyltetrahydrofuran 2 can be formed from (2R,5S)-5-
bromo-2-hexanol 3. [Cf. #3]. What stereoisomer of 3 leads to the enantiomer of 2 [ent-
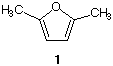
2]? [Structure 1 is 2,5-dimethylfuran].

9. Methyl t-butyl ether (MTBE) is a gasoline additive. How can it be prepared from 2-chloro-
2-methylpropane?
10. MTBE is made industrially from 2-methylpropene (isobutylene) using an acid catalyst instead
of an alkyl halide (see #9). Write a mechanism for this reaction. What is the common intermediate in #9
and #10?