- Chem 220 - Organic
Chemistry
Problem Set 4, Solution
Set
Stereochemistry, Chapter
5
Due: Monday, October
5, 2009

|
The
Borremean Rings
Versions of this symbol date to the time
of the Vikings.
In the 15th century, it was the symbol of a tripartite
alliance of the Milanese families Visconti, Sforza and
Borromeo via intermarriage. Break any (wedding?) ring and
the others separate, hence the alliance is broken. The rings
form a chiral
object (left) that is not
superimposable on its mirror
image. A set of Borremean rings
has been used as the logo for a certain refreshment that
extols purity, body, and flavor. Is the sense of chirality
of the two sets of Borremean rings the same or different?
For some other discourses on chirality, see:
Potpourri
The Figure 8
Knot
Gentlemen's
Neckties
Molecular
Knots
|

|
Read the stereoisomers
module in the StudyAids and do the
exercises. There is no need to record answers on your
homework.
Don't forget the Chirality
of Shells (Powerpoint). Do
left-handed whelks have a better survival rate than their mirror
image brethren? Click here.
|
1. When
(R)-1-chloro-2-methylbutane undergoes free radical
chlorination, four monochloro constitutional isomers are
formed. What are these structures? Draw them. Be explicit as
to diastereomers, enantiomers, racemates, etc.
|
|
2. There are twelve possible Fischer
projections for a given enantiomer of α-chloropropionic
acid. Six of them are shown below. Assign
R,S-configurationns to each one. Draw the remaining
six projections with their R, S-designations.
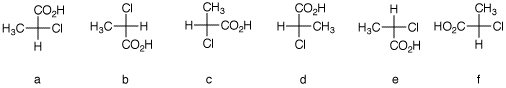
The configurations of
a-f are shown. Row 1 is illustrates what the Fischer
projections mean. [See the end of the answer.]
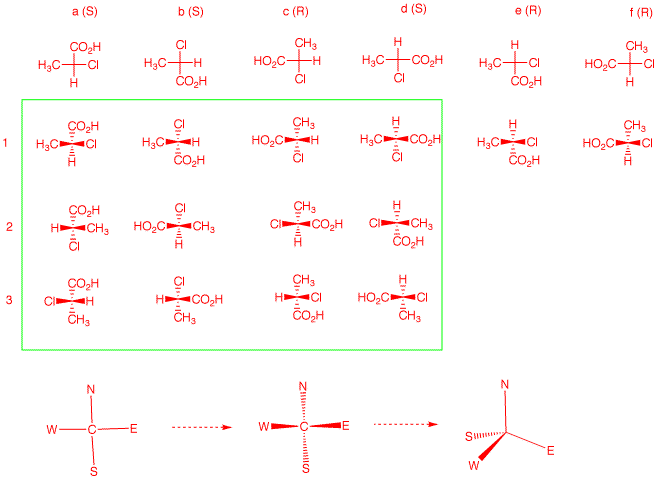
Consider the planar Fischer projection as a compass, NEWS.
Convert the compass to a Fischer projection. If you think of
N being in the rear, then W --> E --> S is clockwise.
Think of the Fischer projection as three-legged stool with
WES as the legs and N as the spindle. Tip the Fischer
projection forward to do this. If you rotate about the C-N
axis, there are three different arrangements of the legs.
but SEW can all occupy the position of N; four choices. For
a given enantiomer of NEWS, there are 12 possible
projections. With in the green box are the 12 projections
for the case at hand. In 1 a-d, four different groups are at
N. Reading down the four columns a-d, one has the three
permutations 1-2-3.
|
3. A 3:1 mixture of
enantiomers has [α]D = -60o. What
is the rotation of the d- and l-enantiomers? Show work.
The major enantiomer is
levorotatory based on the net negative rotation. The mixture is 3/4
levo (nb) and 1/4 dextro (na).
Thus,
obs.
rotation = na (x) + nb (-x) or,
[na and nb are the mole fractions;
na + nb = 1]
-60
= 1/4 (x) + 3/4 (-x)
-60
= -x/2
x
= [α]D = -120 for the levo enantiomer; [α]D
= +120 for the dextro enantiomer.
|
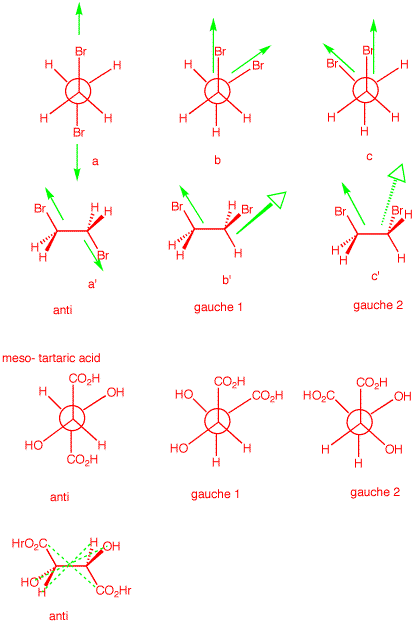
4. a) 1,2-Dibromoethane is optically
inactive yet it has a dipole moment. Explain and illustrate.
[Hint: Draw the staggered conformations and assess
optical activity and dipole moment for each.]
b) meso-Tartaric acid exists in three staggered
conformations, none of which has a plane
of symmetry. Yet the compound is
optically-inactive. Indeed, the only conformation that has a
plane of symmetry is quite unstable. Explain and
illustrate.
|
|
Optical activity is an
algebraically additive property; dipole moments are
cumulative in nature. The three staggered conformations of
1,2-dibromobutane are shown on the right in both Newman
projections and sawhorse views. The anti conformation has no
net dipole. The bond dipoles cancel. [Only the
green
C-Br bond dipoles are shown.] The two gauche
conformations have net dipoles. The vector sum of the bond
dipoles gives the molecular dipole. The anti conformation is
achiral [plane of symmetry]. The gauche
conformations are chiral and form a racemate. no net optical
activity.
|
|
|
5. Which of the following compounds are,
in principle, capable of resolution? Explain and illustrate.
[For 3-D Jmol views of these structures click here.:
5a,
5b,
5c,
5d,
5e,
5f.]

5a) This biphenyl is
not planar owing to the four large groups that inhibit
planarity. The two rings are orthogonal to one another
thereby producing two non-superimposable mirror
images.
b) This compound can
be resolved. Imagine that the CO2H group is above
the plane of the molecule. Draw its mirror image. They are
not superimposable. The doublebond and the 6-membered ring
are an extension of the cumulated double bonds in the
resolvable allenes.
c) Not resolvable.
Free rotation about the biphenyl bond is too rapid for
resolution.
d) This is an
extension of 5b. It is resolvable. An even nummber of
contiguous double bonds or 6-membered rings the compound is
resolvable; odd number, not resolvable.
e) Resolvable. Same as
5a.
f) Eight
CH2 groups in a row are just enough to span the
aromatic ring. Neither the CO2H nor the methyl
group can pass through the large ring. The compound is
resolvable.
|
|
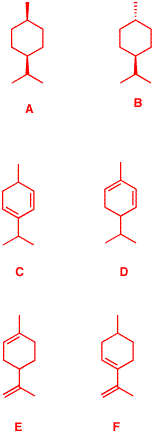
6. (S)-α-Phellandrene
([α]D = +86o) is a
monoterpene with the characteristic fragrance of dill.
(S)-α-Phellandrene reacts with 2 moles of
hydrogen gas in the presence of Pd to give two cyclohexanes
A and B, both of which have the formula
C10H20 and both of which are optically
inactive. Compound A has an energy difference of 0.4
kcal/mol between its two chair conformations while compound
B has a value of 3.8 kcal/mol for the same
equilibrium.
Explain the loss of optical activity, the energy
differences, and identify the structures A and
B.
|
|
6) You can look up the
structure of (S)-α-phellandrene. That two equivalents
of hydrogen are absorbed means that there are two double
bonds. [One triple bond is a possibility but there is no
where to place it.] At least one of the double bonds
must be in the ring and trisubstituted including either the
methyl or isopropyl group. This is true because two
cyclohexanes are formed on reduction. The group not involved
in the trisubstituted double bond is the center of
asymmetry. You can assume that the methyl and the isopropyl
group are 1,4 to one another because A and B are both
achiral [Need a plane of symmetry.] A must be
1,4-cis because it has the smaller energy difference. What
are two numbers whose difference is 0.4 and whose sum is
3.8.
x + y =
3.8
x - y =
0.4
2x =
4.2
x = 2.1 (isopropyl); y
= 1.7 (methyl)
Given the information
in the problem, there are only 4 possible structures for
?-phellandrene: C, D, E and F.
|
|
|
7. (R)-α-Phellandrene has
been reported to have a specific optical rotation of
-217o. This observation suggests that the sample
of the enantiomer used in problem 6 above is contaminated.
Assume that the contaminant is the (R)-enantiomer and
that the (R)-enantiomer
is pure. What percentage of each enantiomer is present in
the sample of problem 6? Show work.
In #3 we solved
for the rotation. Here we calculate the % of each
enantiomer. Pure (R)-(-) = [α]D =
-217o; pure (S)-(+) = [α]D
= +217o; impure (S) = [α]D
= +68o.
obs. rotation = n+ (rot +) + n- (rot
-). Since n+ + n- = 1, then
n- = 1 - n+, or
obs. rotation =
n+ (rot+) + (1-n+)
(rot-), or n+ (rot+) -
(n+-1) (rot+), then factoring
obs. rotation =
(rot+)(2n+- 1), or
obs. rotation/(rot+) =
(2n+- 1) = ee (enantiomeric excess) = op (optical
purity)
op = ee = 68/217 =
0.31
Solving for
n+:
n+ = (1 +
.31)/2 = 0.655, then
n- = 1-
0.655 = 0.345.
(S)-Phellandrene = 65.5%;
(R)-Phellandrene = 34.5%
|





