|
1. Read Degree
(Elements) of Unsaturation. How
many degrees of unsaturation are present in
C8H3BrClN3O2?
Draw a structure that has the number of degrees of
unsaturation you determined and that is necessarily in
agreement with the formula. |
Paul Sabatier 1912 Co-Nobel Prize in Chemistry Hydrogenation by Metal Catalysis |
|
4. Compound A (C5H11Cl) reacts readily with water to form B, C5H12O. Exposure of compound A to aq. NaOH gives only C (major) and D (minor). Hydrogenation of C liberates 27.0±0.1 kcal/mol heat while D liberates 28.5 kcal/mol of heat during hydrogenation. What are the structures A-D? Explain. |
|
5. a) Determine the heat of hydrogenation of cyclohexene from the heat of formation tables. b) How does this value compare with the heat of hydrogenation of an unstrained cis-disubstituted double bond? c) Given the heat of hydrogenation of cyclopentene (chapter 7) determine the heat of formation of cyclopentene. d) BONUS: Why is the heat of hydrogenation less for cyclopentene than that for cyclohexene? Show all work. |
|
6. Two stereoisomers, A and
B, absorb one equivalent of hydrogen upon catalytic
hydrogenation to form cyclooctane. Compound A, which
is capable of resolution, liberates 34.5 kcal/mol of heat
while B liberates 24.3 kcal/mol of heat. |
|
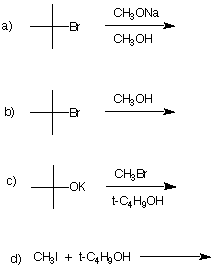
7. Comment critically on the following
proposed synthesis of the now banned gasoline additive,
methyl tertiary-butyl ether (MTBE). If you believe the
reaction will be successful, provide the type of mechanism
that is operable and illustrate it with the curved arrow
formalism. If you feel that the reaction will not be
successful, state the expected product of the reaction and
the mechanism by which it is formed. Illustrate with curved
arrows. If no reaction takes place, state so and explain why
not. |