|
|
|
|
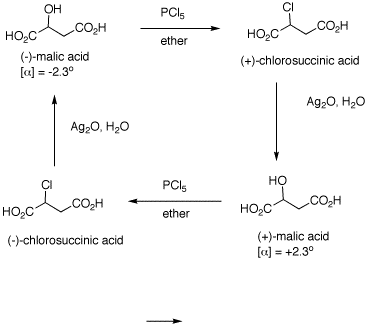
1. The inversion of configuration in an SN2 reaction is often called a Walden inversion, named after its discoverer, Paul Walden. In the cycle shown above, the overall conversion of one enantiomer of malic acid to the other one must require an inversion of configuration. Similarly, the same is true of the chloro acids. More generally, each interconversion of enantiomers must require an odd number of inversions. The PCl5 reaction requires a single inversion which means that the Ag2O reaction involves an even number of inversions of configuration, namely two in this instance. (-)-Malic acid is of the (S)-configuration. a) Show how malic acid, like any alcohol, might react with PCl5 and then undergo inversion to form a chloride. Remember that phosphoric acid is a strong acid and its conjugate base and analogs thereof are also good leaving groups. b) Silver oxide is an anhydrous form of AgOH. The carboxylic acid group closest to the hydroxyl group plays a role in the process. The reaction medium is mildly alkaline. c) Draw these four enantiomers as Fischer projections. (-)-Malic acid is of the (S)-configuration. |
|
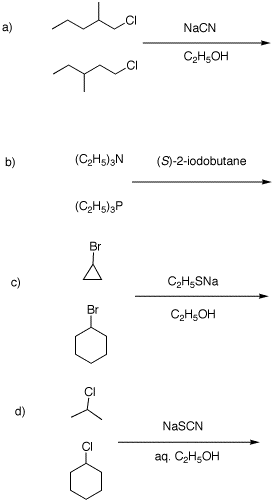
2. In each of the following reactions one equivalent of each reactant competes for one equivalent of reagent (over the arrow) in an SN2 reaction. Only half of the reactants can react because of the limited amount of reagent (two equivalents would be required for complete reaction). Draw the structure of the major product in each example and explain why it is formed preferentially. Provide mechanisms (curved arrow formalism).
|
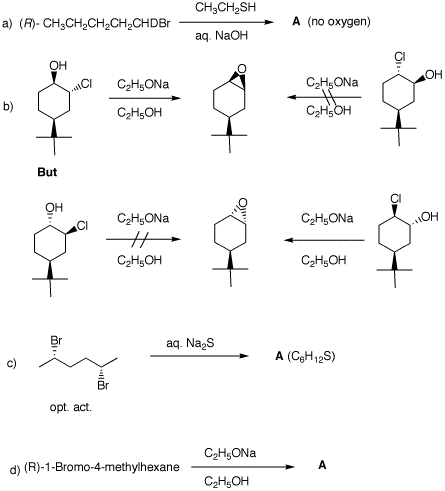
3. A student needs a sample of (S)-2-octanethiol (A). She locates a bottle of (S)-(+)-octanol (B). Unaware of any procedure to transmute oxygen into sulfur nor aware of any procedure that directly substitutes sulfur for oxygen with retention of configuration, she launches a plan of her own. She prepares the tosylate C of the alcohol B. When the tosylate was mixed with excess anhydrous NaI in acetone, iodide D was isolated within a short period of time. Iodide D was allowed to react with excess Na2S, which formed (S)-A with an optical rotation of [α]D= +29.6o.
|
a) Does the formation of tosylate C from alcohol B involve an inversion of configuration? Explain. b) How might tosylate C be formed from tosyl chloride 1? Provide a mechanism. |
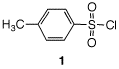 |
c) Using Table 6.4, explain why iodides are formed from tosylates and not vice versa.