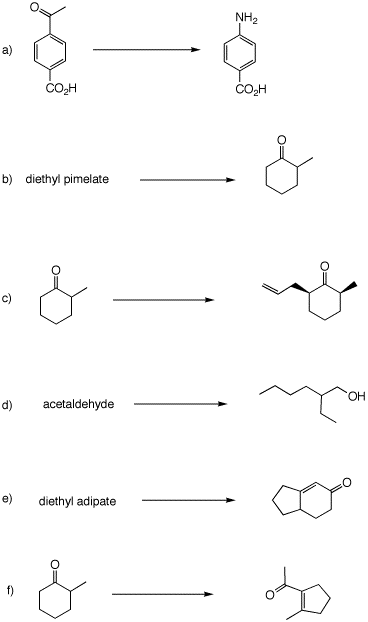
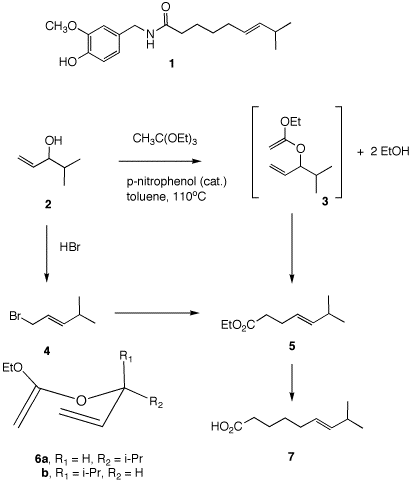
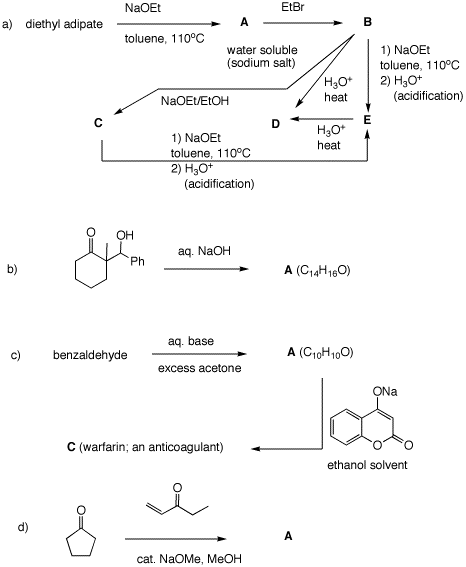
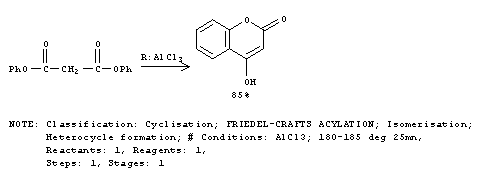
Problem Set 8, Chapter 22
Due: April 11, 2005
Enols and Enolates
The base-mediated condensation of esters
to form β-ketoesters has had an interesting history.
Although the reaction often bears the name of
Ludwig
Claisen (1853-1930), who explored
the scope of this reaction (1887) and related ones, he did
not discover it. In 1863, Geuther discovered that sodium
reacts with ethyl acetate to liberate hydrogen and generate
a new compound C6H9NaO3.
Acidification of the sodium salt afforded "ethyl diacetic
acid". The sodium compound reacted with alkyl halides to
form a series of alkyl ethers. Consequently, Geuther
formulated ethyl diacetic acid as having the the enolic
structure
CH3CH(OH)=CHCO2C2H5. Claisen found vindication in his
mechanism because it explained why diethyl oxalate
successfully condensed with ethyl butyrate but not with
ethyl isobutyrate. Simply put, ethyl isobutyrate only had
one hydrogen available and not the two minimally required by
his mechanism. Dieckmann (1900) demonstrated that the
problem was not that ethyl isobutyrate had only one hydrogen
available but rather that the product formed with diethyl
oxalate was formed reversibly. In subsequent papers, Claisen
would expand the scope of these condensation reactions.
At about the same time, Frankland (of valence
fame) and Duppa (1866) produced the same sodium salt by the
same method. They proposed not only the formation of the the
monosodium salt of ethyl acetate but also its disodium salt
because they were able to isolate products of dialkylation
on carbon. The formulation of Geuther's ethyl diacetic acid
as a ketonic substance by Frankland led to acceptance of his
views and a name change to acetoacetic ester. [You can
see their paper here.
Notice that they drew structures in the form of the then
prevalent type theory. In this paper the Gay-Lussac method
for determining the vapor density of a volatile compound was
employed. A translation of Biot's description of the method
and original diagrams of the apparatus are here.]
By 1877, Wislicenus demonstrated that there were no dianions
in the process and that the dialkylation process was
stepwise. In Claisen's 1887 paper he was able to show that
sodium ethoxide and not sodium was the true condensing
agent. Moreover, he claimed that the same sodio orthoester,
PhC(OCH2Ph)(OCH3)(ONa), was formed
from benzyl benzoate and sodium methoxide or methyl benzoate
and sodium benzylate. He proposed the following "mechanism",
albeit from the "lasso school", for the formation of the
sodium salt of acetoacetic ester.
Nef (1897) suggested that Claisen's sodio orthoester could
lose ethanol to form what we would call the enolate of ethyl
acetate. This species could then react, in some unspecified
way, with ethyl acetate to lead to the sodium salt of
acetoacetic ester. On the other hand, Michael correctly
pointed out that there was no evidence that Claisen's sodio
orthoester even existed. Lapworth (1902) formulated the
correct mechanism for the acetoacetic ester condensation.
The controversy over whether acetoacetic ester existed in
the keto or enol form led to the concept of tautomerism. In
fact, acetoacetic acid exists in both forms. The tautomeric
structures are readily seen in the 13C NMR
spectrum of acetoacetic
ester. Knorr (1911) separated the
enol and keto forms of acetoacetic ester. Source: J. B.
Cohen, Organic Chemistry (1907)