|
1. Study #2 and #3 in the Alkyl Halide module and #1 in the Ether module in ORGO. 2. Hydrocarbon A forms only two
monochlorinated compounds B and C in nearly
equal amounts as predicted by calculation. B is a
racemate and C is achiral. Compound B reacts
with aqueous NaOH to give alcohol D and compound
E. C reacts with water to give alcohol
F and compound G. Both E and G
react with hydrogen and a catalyst to give a saturated
hydrocarbon with a heat
of formation of -42.5 kcal/mol.
What are the structures A-G? Explain and
illustrate. |

Biot examining Pasteur's tartrate crystals
|
|
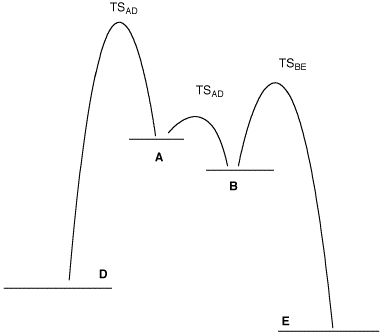
b) A student recognizes that ΔGo is not quite ΔHo but at 400oK, ΔGo=-1.84log10Keq. The difference in the heats of formation of the two geometrical stereoisomers translates into a calculated ratio of 3.5:1. Is he using the correct analysis in spite of the fact that his ratio is not the observed ratio? Explain. c) A and B represent two relevant, staggered conformations of 2-bromobutane. Assign A and B as Newman projections. Explain your choice. d) Why is the third staggered conformation C not included on the diagram? e) What are the two fastest processes in this diagram: A-->B, B-->E, E-->B, B-->A, A-->D, or D-->A? Explain. f) Another student suggests that the observed ratio is determined by the difference in the rate of formation of D vs. E. Show how this difference is reflected in the diagram. |
 |
|
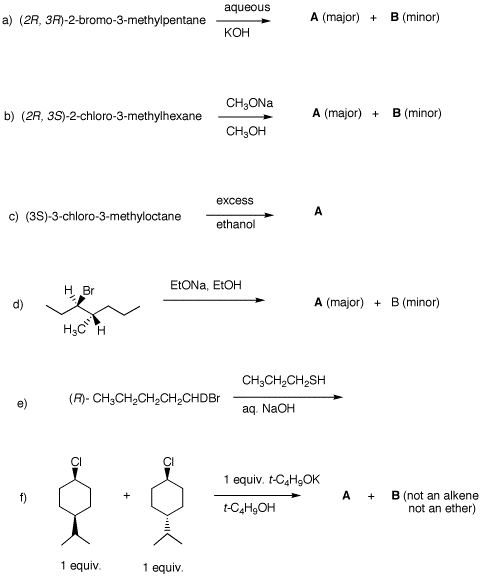
4. Provide the products in each of the following questions and mechanisms for their formation. Pay attention to stereochemistry. Be explicit about enantiomers.
|
|
5. Compound A reacts very slowly with hot KOH to form alcohol B. No products of elimination are formed. When A is heated with aqueous AgNO3, AgBr is formed in addition to C (C5H12O), D (major) and E (minor). D and E absorb 1 equiv. of H2 in the presence of a Pd catalyst to form 2-methylbutane. What are the structures of A-E? Explain and illustrate. |
|
6. Compound A, (1S, 2R, 5S)-1-chloro-2-isopropyl-5-methylcyclohexane, and compound B, (1S, 2S, 5R)-1-chloro-2-isopropyl-5-methylcyclohexane both react with C2H5ONa in ethanol to give products of elimination. A gives only C while B gives a 1:3 ratio of D and E, respectively. Compounds C, D, and E are reduced with H2 to give cyclohexanes. Reduction of C or D gives F, while reduction of E affords F and G. a) What are the structures A-G? Explain and illustrate. Provide clear mechanisms for the formation of C-E. b) What is the relationship between C and D? c) Which compounds are optically active? Which ones are not? d) Imagine that an equimolar mixture of A and B reacts with half as much sodium ethoxide. Does a A or B react faster? Why? What is formed? |