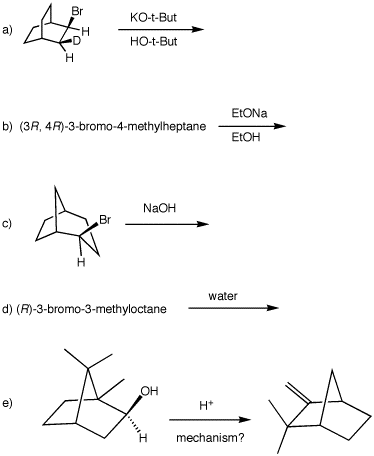
|
1. Read Degrees (Elements) of
Unsaturation here
and/or here.
How many degrees of unsaturation are present in
C4H5BrClNO2? Draw a
structure that has the number of degrees of unsaturation you
determined and that is necessarily in agreement with the
formula. a) How does your answer compare with the typical value reported in Table 7-1 on pg. 285? Explain. b) Find the heat of formation of cyclooctane. Why is its value nearly the same as that of cyclohexane and not ~-40 kcal/mol? c) The heat of hydrogenation of cis-cyclooctene is -24.3 kcal/mol. What is its heat of formation? Show work. d) What accounts for the difference in the heats of hydrogenation of cyclohexene and cis-cyclooctene? e) The heat of hydrogenation of trans-cyclooctene is -34.5 kcal/mol. What is the heat of isomerization of the geometrical stereoisomers of cyclooctene and in what direction does it occur? Place the data above in a chart that relates all of the the heats relative to the steady state. |
Paul Sabatier 1912 Co-Nobel Prize in Chemistry Hydrogenation by Metal Catalysis |
|
4. Optically active alkyl chloride A (C6H13Cl) is converted into a mixture of two hydrocarbons B and C (major) upon exposure to CH3ONa/CH3OH. Compound (R)-B liberates 30.2 kcal/mol of heat upon hydrogenation while compound C liberates 26.2 kcal/mol. A stereoisomer of A, namely D, when treated with CH3ONa/CH3OH affords (S)-B and E. Compound E liberates 25.8 kcal/mol of heat upon hydrogenation. The hydrogenation product F of B, C, and E is achiral. Free radical bromination of F gives monobromide G, which, when treated with CH3ONa/CH3OH, gives C, E, and H, all three of which have the same formula. What are the structures and IUPAC names of A-H? Explain and illustrate. |
|
5. Achiral compound A (C9H16) is inert toward hydrogenation. Free radical chlorination of A provides four (theoretically) monochloro compounds: primary achiral B (11%), secondary (±)-C and (±)-D in 68% combined yield, and tertiary achiral E (21%). There is much less D formed than C even though they arise from the same carbon radical. Compound A reacts reluctantly with NaOH at elevated temperatures to give F (C9H18O). No products of elimination are formed. Upon exposure to NaOH, compounds C and D give G (C9H16). Compound E is inert toward NaOH. a) What are the structures A through G? Explain. b) Why is less D formed than C? Explain. |
|
6. Complete each of the following questions. Provide brief explanations.
|