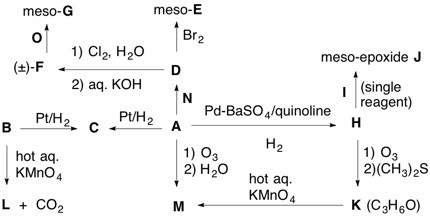
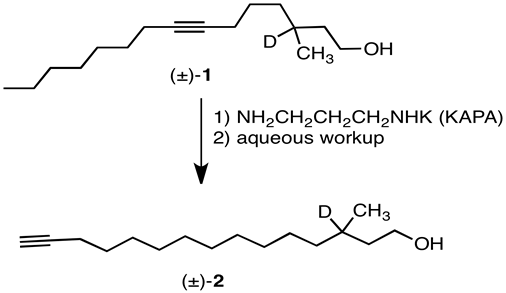
Due: April 8, 2013

Friedrich Wöhler (1800-1884) (Wöhler possessed a wry sense of humor) |
Aluminum was once a precious metal although it was plentiful. The problem was how to remove it from its ore. Friedrich Wöhler, of urea synthesis fame, was able to accomplish this feat but by an impractical method. He was to meet a young chemist, Frank Jewett, recently arrived in Göttingen from Yale. Aware of the difficulty Wöhler had had and probably encouraged by Wöhler, Jewett, as a professor at Oberlin College, passed the problem onto Charles Martin Hall, a young student at the college. Hall solved the problem in his family garage. Thus was born Alcoa. At the same time in Spray, North Carolina, Thomas Willson, a Canadian, and American James Moorhead were unsuccessfully trying to refine aluminum using an electric arc. Unsuccessful in purifying aluminum, they sought calcium metal. Heating coal tar and lime in an electric furnace they obtained a brittle material that produced a combustible gas upon exposure to water. The material was not calcium nor was the gas hydrogen. The pair was calcium carbide and acetylene, the basis for Union Carbide Corporation (RIP). |

Charles Martin Hall (1863-1914) |
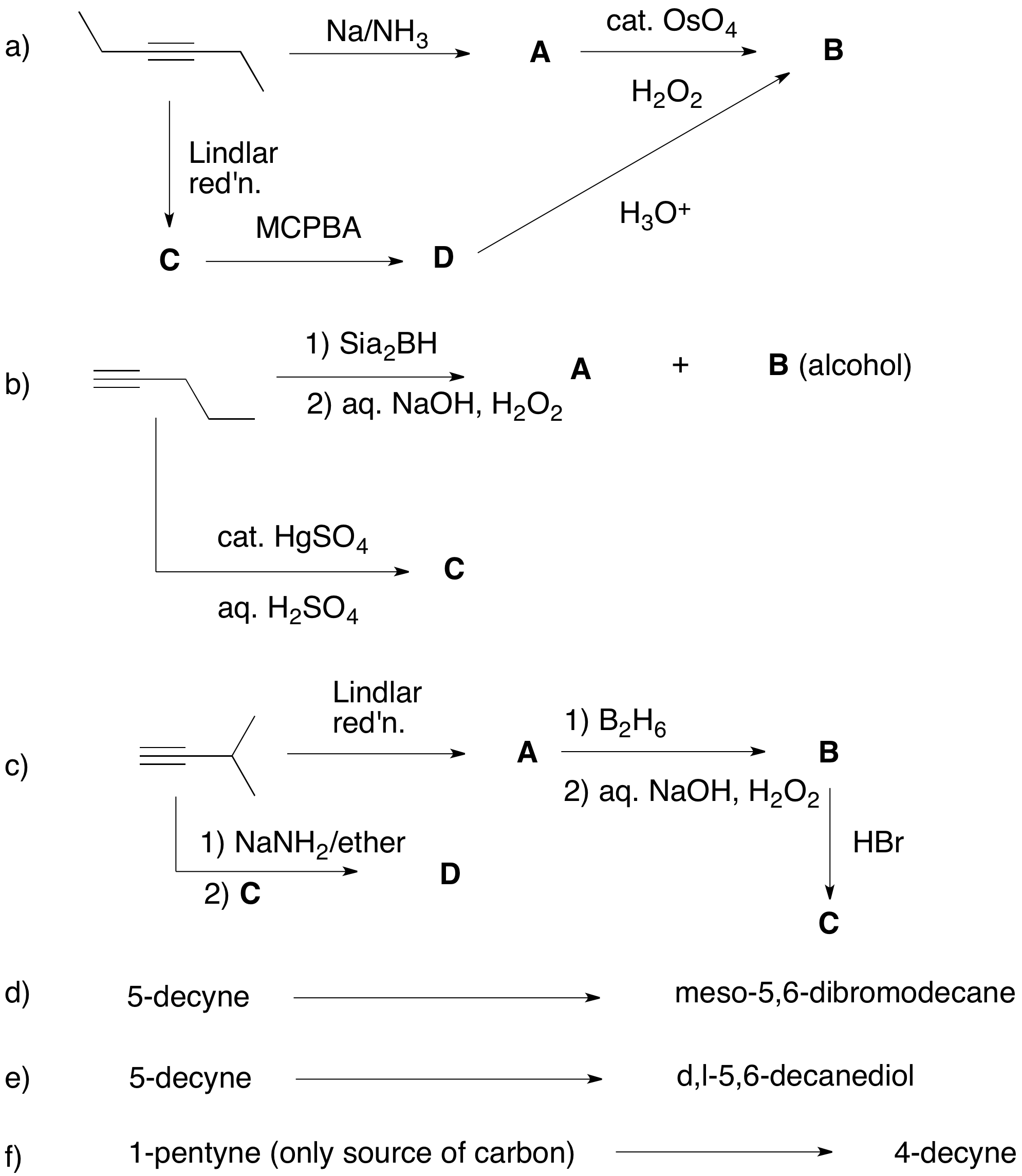 .
.