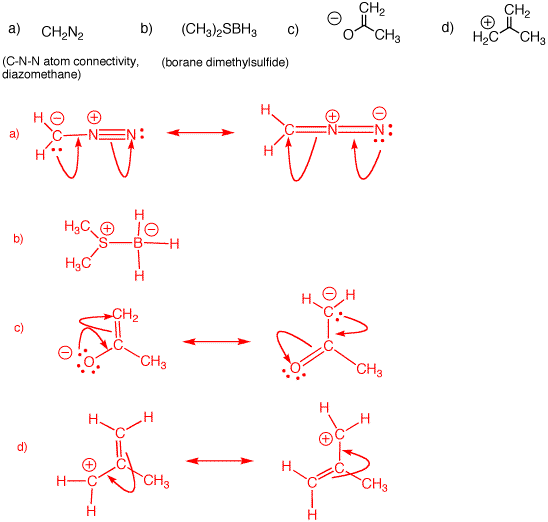
Problem Set 1, Solution Set
Chapters 1 and 2, Structure, Bonding, Reactivity
Due: Monday, September 13, 2010
John Dalton (1766-1844) John Dalton's formulation of an
Atomic
Theory in the first decade of the
19th century provided a theoretical basis for understanding
chemical behavior. In addition to defining the Law of
Multiple Proportions, he also formulated the Rule of
Greatest Simplicity, which held that water was a binary
compound, OH. (Note: Dalton did not use our modern symbols,
which came to us from Berzelius,
but rather
circles that were distinguishable
from one another.) Dalton established the combining masses
of H to O in water as ~1:6. This ratio was later refined to
1:8. Dalton postulated that in a molecules comprised of two
different atoms, the simplest one in the series would be
binary. While this rule applied to CO and CO2, it
did not apply to the pair, water and hydrogen peroxide.
Thus, water, according to Dalton, was OH. The
Rule
of Greatest Simplicity, which was
at odds with Gay-Lussac's
Law of Combining Volumes of Gases that demonstrated the
volume of hydrogen produced upon electrolysis of water was
twice that of oxygen, was dismissed by Dalton as a faulty
result. Moreover, although there was agreement regarding the
combining masses of atoms in the first half of the
nineteenth century, there was
disagreement as to the unit mass
of the common atoms encountered in organic chemistry:
hydrogen (1), carbon
(2x6 or 1x12), oxygen (2x8 or
1x16). Since hydrogen was the lightest of the elements, it
was assigned a mass of one (Prout's
Hypothesis), a notion that is
unrelated to today's mass of hydrogen owing to the presence
of a single proton in the hydrogen nucleus. Berzelius's
proposal of a mass scale based upon O = 100 would have
worked as well. For a Brief History of Organic Chemistry
(PowerPoint), click
here.
1. The chemical structures shown
below all occur in nature. They have also been made
(synthesized) by chemical means from simpler organic
compounds in this department over the past 40 years.
[See
the background on the website homepage.] You will learn
about Classes of Compounds one class at a time. They will be
for the most part mono-functional compounds. All of the
compounds shown below are multi-functional
compounds. a) Identify the Class of Compound of the
functionality present with in each of the circles. Print
this page and use it to designate answers. [See the
inside front cover of your textbook for Classes of
Compounds, Functional Groups and Abbreviations.]
See answers in the
diagram below (red italics). The compounds that end in -one
contain ketones while the compounds ending in -ol contain
alcohols. This is a common way of naming compounds but not
necessarily. b) You should have identified two
alcohols [ROH, where R = alkyl (aliphatic), not
aryl (aromatic)]. Of these two alcohols, one is said to
be primary, the other tertiary. Why? Is there another
primary alcohol in the structures? Another tertiary alcohol?
Are there any secondary alcohols?
Cyclophellitol has
a primary alcohol circled. The carbon to which the hydroxyl
group is attached has one (primary) carbon attached to it
while the circled alcohol in steviol has a carbon bearing
one hydroxyl group and three (tertiary) carbons. [The
definition need not be only attached carbons. The compound
CH3OCH2Cl, is a primary chloride even
though there are no carbons attached to the carbon to which
the chlorine is attached.] The hydroxyl group in the
center of forskolin is tertiary. There are five secondary
alcohols: three in cyclophellitol and two in forskolin. The
symbol Ac in forskolin stands for acetyl (see the Tables on
the inside cover of your text). Acetic acid may be written
in shorthand as AcOH.