Read the stereoisomers
module in the StudyAids and do the
exercises. There is no need to record answers on your
homework.
Don't forget the Chirality
of Shells (Powerpoint). Do
left-handed whelks have a better survival rate than their mirror
image brethren? Click here.
|
1. When
(R)-1-chloro-2-methylbutane undergoes free radical
chlorination, four monochloro constitutional isomers are
formed. What are these structures? Draw them. Be explicit as
to diastereomers, enantiomers, racemates, etc.
|
|
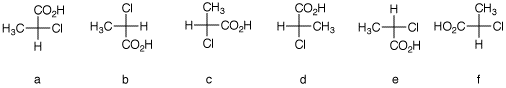
2. There are twelve possible Fischer
projections for a given enantiomer of α-chloropropionic
acid. Six of them are shown below. Assign
R,S-configurationns to each one. Draw the remaining
six projections with their R, S-designations.
|
3. A 3:1 mixture of
enantiomers has [α]D = -60o. What
is the rotation of the d- and l-enantiomers? Show
work.
|
4. a) 1,2-Dibromoethane is optically
inactive yet it has a dipole moment. Explain and illustrate.
[Hint: Draw the staggered conformations and assess
optical activity and dipole moment for each.]
b) meso-Tartaric acid exists in three staggered
conformations, none of which has a plane
of symmetry. Yet the compound is
optically-inactive. Indeed, the only conformation that has a
plane of symmetry is quite unstable. Explain and
illustrate.
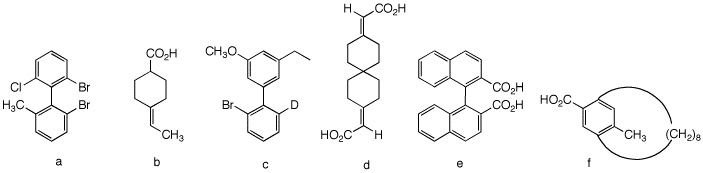
5. Which of the compounds on the right are, in principle,
capable of resolution? Explain and illustrate. [For 3-D
Jmol views of these structures click here.: 5a,
5b,
5c,
5d,
5e,
5f.]
6. (S)-α-Phellandrene
([α]D = +86o) is a
monoterpene with the characteristic fragrance of dill.
(S)-α-Phellandrene reacts with 2 moles of
hydrogen gas in the presence of Pd to give two cyclohexanes
A and B, both of which have the formula
C10H20 and both of which are optically
inactive. Compound A has an energy difference of 0.4
kcal/mol between its two chair conformations while compound
B has a value of 3.8 kcal/mol for the same
equilibrium.
Explain the loss of optical activity, the energy
differences, and identify the structures A and
B.
|
|
|
7. (R)-α-Phellandrene has
been reported to have a specific optical rotation of
-217o. This observation suggests that the sample
of the enantiomer used in problem 6 above is contaminated.
Assume that the contaminant is the (R)-enantiomer and
that the (R)-enantiomer
is pure. What percentage of each enantiomer is present in
the sample of problem 6? Show work.
|


